Impact Factor
Theranostics 2018; 8(17):4750-4764. doi:10.7150/thno.26108 This issue Cite
Research Paper
Optogenetic sensors in the zebrafish heart: a novel in vivo electrophysiological tool to study cardiac arrhythmogenesis
1. Department of Medical Physiology, Division of Heart & Lungs, University Medical Center Utrecht, Yalelaan 50, 3584 CM, Utrecht, The Netherlands.
2. Hubrecht Institute, Royal Netherlands Academy of Arts and Sciences (KNAW), University Medical Centre Utrecht, 3584 CT, Utrecht, The Netherlands.
3. Laboratory for Neuronal Circuit Dynamics, Imperial College London, London, UK
4. Centre for Neurotechnology, Institute of Biomedical Engineering, Imperial College London, London, UK.
5. Champalimaud Research, Champalimaud Centre for the Unknown, Lisbon, Portugal
* these authors contributed equally
Abstract
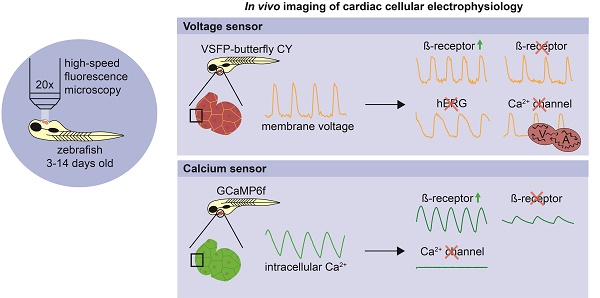
Cardiac arrhythmias are among the most challenging human disorders to diagnose and treat due to their complex underlying pathophysiology. Suitable experimental animal models are needed to study the mechanisms causative for cardiac arrhythmogenesis. To enable in vivo analysis of cardiac cellular electrophysiology with a high spatial and temporal resolution, we generated and carefully validated two zebrafish models, one expressing an optogenetic voltage indicator (chimeric VSFP-butterfly CY) and the other a genetically encoded calcium indicator (GCaMP6f) in the heart.
Methods: High-speed epifluorescence microscopy was used to image chimeric VSFP-butterfly CY and GCaMP6f in the embryonic zebrafish heart, providing information about the spatiotemporal patterning of electrical activation, action potential configuration and intracellular Ca2+ dynamics. Plotting VSFP or GCaMP6f signals on a line along the myocardial wall over time facilitated the visualization and analysis of electrical impulse propagation throughout the heart. Administration of drugs targeting the sympathetic nervous system or cardiac ion channels was used to validate sensitivity and kinetics of both zebrafish sensor lines. Using the same microscope setup, we imaged transparent juvenile casper fish expressing GCaMP6f, demonstrating the feasibility of imaging cardiac optogenetic sensors at later stages of development.
Results: Isoproterenol slightly increased heart rate, diastolic Ca2+ levels and Ca2+ transient amplitudes, whereas propranolol caused a profound decrease in heart rate and Ca2+ transient parameters in VSFP-Butterfly and GCaMP6f embryonic fish. Ikr blocker E-4031 decreased heart rate and increased action potential duration in VSFP-Butterfly fish. ICa,L blocker nifedipine caused total blockade of Ca2+ transients in GCaMP6f fish and a reduced heart rate, altered ventricular action potential duration and disrupted atrial-ventricular electrical conduction in VSFP-Butterfly fish. Imaging of juvenile animals demonstrated the possibility of employing an older zebrafish model for in vivo cardiac electrophysiology studies. We observed differences in atrial and ventricular Ca2+ recovery dynamics between 3 dpf and 14 dpf casper fish, but not in Ca2+ upstroke dynamics.
Conclusion: By introducing the optogenetic sensors chimeric VSFP-butterfly CY and GCaMP6f into the zebrafish we successfully generated an in vivo cellular electrophysiological readout tool for the zebrafish heart. Complementary use of both sensor lines demonstrated the ability to study heart rate, cardiac action potential configuration, spatiotemporal patterning of electrical activation and intracellular Ca2+ homeostasis in embryonic zebrafish. In addition, we demonstrated the first successful use of an optogenetic sensor to study cardiac function in older zebrafish. These models present a promising new research tool to study the underlying mechanisms of cardiac arrhythmogenesis.
Keywords: Optogenetics, zebrafish, in vivo cardiac cellular electrophysiology, chimeric VSFP-butterfly CY, GCaMP6f
 Global reach, higher impact
Global reach, higher impact