13.3
Impact Factor
Theranostics 2018; 8(17):4824-4836. doi:10.7150/thno.24747 This issue Cite
Research Paper
Neurogliovascular dysfunction in a model of repeated traumatic brain injury
1. University of Toronto, Department of Medical Biophysics, Toronto, Ontario, Canada
2. Sunnybrook Research Institute, Toronto, Ontario, Canada
3. Fundamental Neurobiology, Krembil Research Institute, University Health Network, M5T 2S8 Toronto, Ontario, Canada
4. Mouse Imaging Centre, The Hospital for Sick Children, Toronto, Ontario, Canada
5. Université Grenoble Alpes, Grenoble Institut des Neurosciences, Grenoble, France
6. Bruker Biospin MRI, Ettlingen, Germany
7. Inserm, U1216, Grenoble, France
8. University of Toronto, Department of Laboratory Medicine and Pathobiology, Toronto, Ontario Canada
Abstract
Traumatic brain injury (TBI) research has focused on moderate to severe injuries as their outcomes are significantly worse than those of a mild TBI (mTBI). However, recent epidemiological evidence has indicated that a series of even mild TBIs greatly increases the risk of neurodegenerative and psychiatric disorders. Neuropathological studies of repeated TBI have identified changes in neuronal ionic concentrations, axonal injury, and cytoskeletal damage as important determinants of later life neurological and mood compromise; yet, there is a paucity of data on the contribution of neurogliovascular dysfunction to the progression of repeated TBI and alterations of brain function in the intervening period.
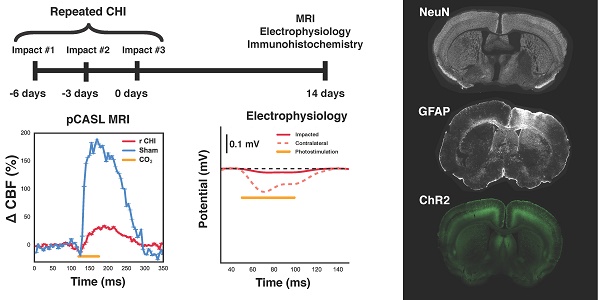
Methods: Here, we established a mouse model of repeated TBI induced via three electromagnetically actuated impacts delivered to the intact skull at three-day intervals and determined the long-term deficits in neurogliovascular functioning in Thy1-ChR2 mice. Two weeks post the third impact, cerebral blood flow and cerebrovascular reactivity were measured with arterial spin labelling magnetic resonance imaging. Neuronal function was investigated through bilateral intracranial electrophysiological responses to optogenetic photostimulation. Vascular density of the site of impacts was measured with in vivo two photon fluorescence microscopy. Pathological analysis of neuronal survival and astrogliosis was performed via NeuN and GFAP immunofluorescence.
Results: Cerebral blood flow and cerebrovascular reactivity were decreased by 50±16% and 70±20%, respectively, in the TBI cohort relative to sham-treated animals. Concomitantly, electrophysiological recordings revealed a 97±1% attenuation in peri-contusional neuronal reactivity relative to sham. Peri-contusional vascular volume was increased by 33±2% relative to sham-treated mice. Pathological analysis of the peri-contusional cortex demonstrated astrogliosis, but no changes in neuronal survival.
Conclusion: This work provides the first in-situ characterization of the long-term deficits of the neurogliovascular unit following repeated TBI. The findings will help guide the development of diagnostic markers as well as therapeutics targeting neurogliovascular dysfunction.
Keywords: repeated TBI, neurovascular, optogenetics, closed head injury
 Global reach, higher impact
Global reach, higher impact