Impact Factor
Theranostics 2019; 9(4):932-944. doi:10.7150/thno.30408 This issue Cite
Research Paper
Quantitative MALDI Imaging of Spatial Distributions and Dynamic Changes of Tetrandrine in Multiple Organs of Rats
1. State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing, 210009, China
2. School of Traditional Chinese Pharmacy, China Pharmaceutical University, Nanjing, 211198, China
* These authors contributed equally.
Abstract
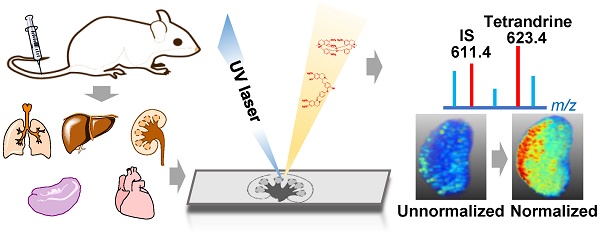
Detailed spatio-temporal information on drug distribution in organs is of paramount importance to assess drug clinically-relevant properties and potential side-effects. Matrix-assisted laser desorption/ionization mass spectrometry imaging (MALDI MSI) as a label-free and sensitive imaging modality provides an additional means of accurately visualizing drug and its metabolites distributions in tissue sections. However, technical limitations, complex physiochemical environment of surface and low abundance of target drugs make quantitative MALDI imaging of drug and its metabolites quite challenging.
Methods: In this study, an internal standard correction strategy was applied for quantitative MALDI imaging of tetrandrine in multiple organs of rats including lung, liver, kidney, spleen, and heart. The feasibility and reliability of the developed quantitative MSI method were validated by conventional liquid chromatography-tandem MS (LC-MS/MS) analysis, and the two methods showed a significant correlation.
Results: The quantitative MALDI imaging method met the requirements of specificity, sensitivity and linearity. Tissue-specific spatio-temporal distribution patterns of tetrandrine in different organs were revealed after intravenous administration in the rat. Moreover, demethylated metabolite was detected in liver tissues.
Conclusions: The current work illustrates that quantitative MALDI imaging provides an alternative means of accurately addressing the problem of drug and its metabolites distribution in tissues, complementary to traditional LC-MS/MS of tissue homogenates and whole-body autoradiography (WBA). Quantitative spatio-chemical information obtained here can improve our understanding of pharmacokinetics (PK), pharmacodynamics (PD), and potential transient toxicities of tetrandrine in organs, and possibly direct further optimization of drug properties to reduce drug-induced organ toxicity.
Keywords: quantitative MALDI imaging, drug tissue distribution, tetrandrine, spatio-temporal heterogeneity of drug metabolism
 Global reach, higher impact
Global reach, higher impact