13.3
Impact Factor
Theranostics 2019; 9(8):2268-2281. doi:10.7150/thno.30649 This issue Cite
Research Paper
Using near-infrared enhanced thermozyme and scFv dual-conjugated Au nanorods for detection and targeted photothermal treatment of Alzheimer's disease
National Engineering Laboratory for AIDS Vaccine, Key Laboratory for Molecular Enzymology and Engineering of Ministry of Education, School of Life Science, Jilin University, Changchun, 130012, China
* These authors contributed equally to this work.
Abstract
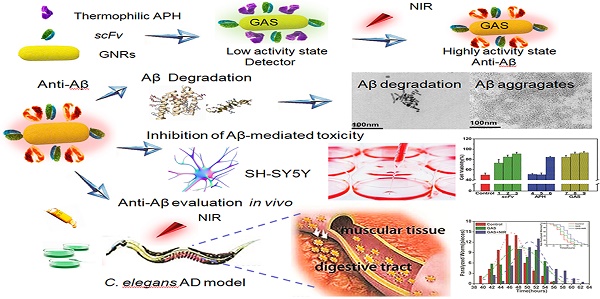
Investigation of targeting inhibitors of Aβ aggregation, heme-Aβ peroxidase-like activity and efficient detectors of Aβ aggregation, are of therapeutic value and diagnostics significance for the treatment of Alzheimer's disease (AD). Due to the complex pathogenesis of AD, theranostics treatment with multiple functions are necessary. Herein we constructed the NIR absorption property of gold nanorods (GNRs) loaded with single chain variable fragment (scFv) 12B4 and thermophilic acylpeptide hydrolase (APH) ST0779 as a smart theranostic complex (GNRs-APH-scFv, GAS), which possesses both rapid detection of Aβ aggregates and NIR photothermal treatment that effectively disassembles Aβ aggregates and inhibits Aβ-mediated toxicity.
Methods: We screened targeting anti-Aβ scFv 12B4 and thermophilic acylpeptide hydrolase as amyloid-degrading enzyme, synthesized GAS gold nanorods complex. The GAS was evalued by Aβ inhibition and disaggregation assays, Aβ detection assays, Aβ mediated toxicity assays in vitro. In vivo, delaying Aβ-induced paralysis in AD model of Caenorhabditis elegans was also tested by GAS.
Results: In vitro, GAS has a synergistic effect to inhibit and disassociate Aβ aggregates, in addition to decrease heme-Aβ peroxidase-like activity. In cultured cells, treatment with GAS reduces Aβ-induced cytotoxicity, while also delaying Aβ-mediated paralysis in CL4176 C.elegans model of AD. Furthermore, the photothermal effect of the GAS upon NIR laser irradiation not only helps disassociate the Aβ aggregates but also boosts APH activity to clear Aβ. The GAS, as a targeting detector and inhibitor, allows real-time detection of Aβ aggregates.
Conclusion: These results firstly highlight the combination of scFv, APH and nanoparticles to be theranostic AD drugs. Taken together, our strategy provides a new thought into the design of smart compounds for use as efficiently therapeutic and preventive agents against AD. Moreover, our design provides broad prospects of biomedical strategy for further theranostics application in those diseases caused by abnormal protein.
 Global reach, higher impact
Global reach, higher impact