Impact Factor
Theranostics 2019; 9(13):3674-3686. doi:10.7150/thno.32943 This issue Cite
Research Paper
Theranostic application of lipiodol for transarterial chemoembolization in a VX2 rabbit liver tumor model
1. Department of Radiology and Biomedical Imaging, Yale School of Medicine, New Haven, Connecticut, USA
2. Department of Radiology, University Medical Center Utrecht, Utrecht, The Netherlands
3. Medical faculty, Utrecht University, Utrecht, The Netherlands
4. Founder, Prescience labs now Cage Pharma, USA
5. Russell H. Morgan Department of Radiology and Radiological Science, Division of Vascular and Interventional Radiology, The Johns Hopkins Hospital, Baltimore, Maryland.
6. Department of Pathology, Yale School of Medicine, New Haven, Connecticut, USA
7. Department of Diagnostic and Interventional Radiology, Lausanne University Hospital, Lausanne, Switzerland
Abstract
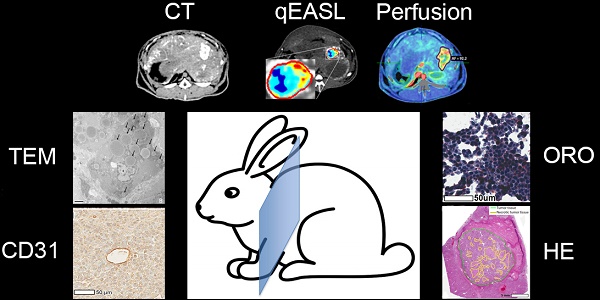
The goal of this study was to investigate the role of Lipiodol as a tumor-specific imaging biomarker to determine therapeutic efficacy of cTACE and investigate its inter-dependency with tumor perfusion using radiological-pathological correlation in an animal model of liver cancer.
Methods: A total of N=36 rabbits were implanted in the left lobe of the liver with VX2 tumors, treated with cTACE using doxorubicin suspended in Lipiodol, and randomly sacrificed at 24 h, 7 days, or 20 days post-TACE. Unenhanced and contrast-enhanced CT scans including a perfusion protocol were obtained before cTACE and immediately before sacrifice. Tumor vascularity and Lipiodol deposition within tumors and hepatic tissue (non-target deposits) were quantified using 3D quantitative assessment tools and measurements of arterial flow, portal flow, and perfusion index (PI). After sacrifice histologic staining, including hematoxylin and eosin (H&E), CD31, and Oil Red O (ORO) were performed on tumor and liver samples to evaluate necrosis, microvascular density (MVD), and Lipiodol retention over time. Transmission electron microscopy (TEM) was performed to assess Lipiodol deposition and clearance over time.
Results: All cTACE procedures were carried out successfully except for one, which was excluded from further analysis. Twenty-four hours post-TACE, tumor PI (p=0.04) was significantly decreased, which was maintained at 7 days (p=0.003), but not at 20 days (p=0.4). A strong correlation (R2 = 0.894) was found between the volume of enhancing tumor tissue at baseline and Lipiodol-positive tumor volume post-TACE. Both ORO and TEM showed deposition of Lipiodol across all imaging time points within the VX2 tumors. However, gradual and ultimately near-complete Lipiodol washout was observed over time in the non-tumoral liver. MVD decreased between 24 h and 7 days post-TACE, and then increased 20 days post-TACE (both p<0.01).
Conclusions: Our data provide radiology-pathology evidence for the function of Lipiodol as a theranostic, tumor-specific drug delivery agent because it is both imageable and tumor-seeking, whereby it is preferentially taken up and retained by tumor cells. Those tumor-specific functions also enable Lipiodol to act as an imaging biomarker for the therapeutic efficacy of cTACE. Together with volumetric quantification of tumor vascularization on CT, Lipiodol could be used as a predictor of a patient's response to cTACE and contribute to the therapeutic management of patients with liver cancer.
Keywords: TACE, Lipiodol, VX2 liver tumor model, HCC, biomarker
 Global reach, higher impact
Global reach, higher impact