13.3
Impact Factor
Theranostics 2019; 9(13):3825-3839. doi:10.7150/thno.33958 This issue Cite
Research Paper
pH- and photothermal-driven multistage delivery nanoplatform for overcoming cancer drug resistance
1. National Engineering Research Center for Nanomedicine, College of Life Science and Technology, Huazhong University of Science and Technology, Wuhan 430074, China
2. Hubei Key Laboratory of Bioinorganic Chemistry and Materia Medica, Huazhong University of Science and Technology, Wuhan 430074, China
*These authors contributed equally to this work.
Abstract
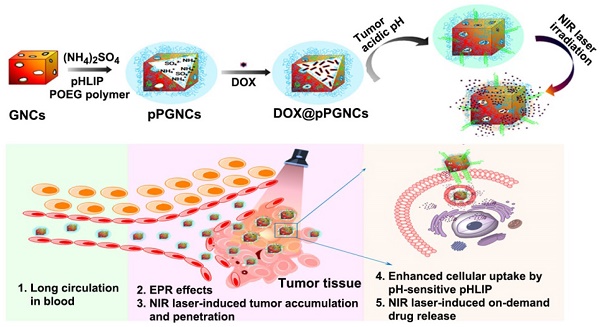
Reversing multidrug resistance (MDR) remains a big challenge in cancer therapy. Combining the hyperthermia and chemotherapy is a promising strategy for efficient cancer treatment with MDR reversal. Gold nanocages (GNCs) are an ideal photothermal (PTT)-chemotherapy integration platform due to their good photothermal conversion efficiency and the unique hollow interiors. However, insufficient tumor cell internalization and in vivo premature drug leakage restrict the anticancer activity of GNCs-based drug delivery systems.
Methods: pH low insertion peptide (pHLIP)- and thermoresponsive poly(di(ethylene glycol) methyl ether methacrylate-co-oligo(ethylene glycol) methyl ether methacrylate) polymer-conjugated GNCs were rationally constructed to load anticancer drug doxorubicin (DOX@pPGNCs). Tumor acidic environment-responsive tumor cell internalization, and near-infrared (NIR) laser-induced tumor accumulation, penetration and on-demand drug release were systematically examined.
Results: DOX@pPGNCs display good photothermal efficacy and thermoresponsive property. NIR laser irradiations at the tumor site significantly enhance tumor accumulation and penetration. Once DOX@pPGNCs reach the tumor site, the conformational transformation of pHLIP at the acidic tumor microenvironment contributes to the enhanced cellular internalization. Furthermore, NIR laser-triggered photothermal effects induce the shrinkage of thermoresponsive polymer, resulting in the opening of the pores of GNCs and a rapid intracellular DOX release to the nuclei. DOX@pPGNCs exhibit synergistic antitumor effect with MDR reversal in vitro and in vivo.
Conclusion: DOX@pPGNCs present strong potential to overcome MDR in cancer.
Keywords: pHLIP, temperature-sensitive polymer, tumor cell targeting, tumor accumulation and penetration, on-demand drug release, multidrug resistance reversal
 Global reach, higher impact
Global reach, higher impact