Impact Factor
Theranostics 2019; 9(13):3966-3979. doi:10.7150/thno.32172 This issue Cite
Research Paper
Engineering temperature-sensitive plateletsomes as a tailored chemotherapy platform in combination with HIFU ablation for cancer treatment
Center for Bioactive Products, Northeast Forestry University/ Key Laboratory of Saline-Alkali Vegetation Ecology Restoration, Ministry of Education, Harbin 150040, China.
Abstract
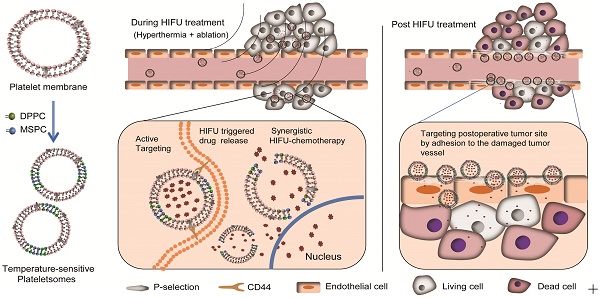
Chemotherapy is widely used in combination with high-intensity focused ultrasound (HIFU) ablation for cancer therapy; however, the spatial and temporal integration of chemotherapy and HIFU ablation remains a challenge. Here, temperature-sensitive plateletsomes (TSPs) composed of platelet (PLT) membrane, 1-stearoyl-2-hydroxy-sn-glycero-3-phosphocholine and 1,2-dipalmitoyl-sn-glycero-3-phosphocholine were developed to adequately integrate chemotherapy with HIFU tumor ablation in vivo.
Methods: The thermosensitive permeability of TSPs was evaluated under both water bath heating and HIFU hyperthermia. The targeting performance, pharmacokinetic behavior and therapeutic potential of TSPs in combination with HIFU ablation were evaluated using HeLa cells and a HeLa cell tumor-bearing nude mouse model in comparison with temperature-sensitive liposomes (TSLs).
Results: TSPs showed high drug loading efficiency and temperature-sensitive permeability. When applied in vivo, TSPs showed a circulation lifetime comparable to that of TSLs and exhibited PLT-specific cancer cell affinity and a vascular damage response. Upon HIFU hyperthermia, TSPs displayed ultrafast drug release and enhanced tumor uptake, providing high drug availability in the tumor site to cooperate with HIFU ablation. After HIFU ablation, TSPs rapidly targeted the postoperative tumor site by adhesion to the damaged tumor vasculature, leading to targeted and localized postoperative chemotherapy.
Conclusion: Due to effective integration at both intraoperative and postoperative stages, TSPs could be a promising chemotherapy nanoplatform in combination with HIFU ablation for cancer therapy.
 Global reach, higher impact
Global reach, higher impact