13.3
Impact Factor
Theranostics 2019; 9(22):6631-6645. doi:10.7150/thno.33353 This issue Cite
Research Paper
Disruption of mitochondrial homeostasis with artemisinin unravels anti-angiogenesis effects via auto-paracrine mechanisms
1. Department of Obstetrics and Gynecology, Kaohsiung Veterans General Hospital, Kaohsiung, Taiwan;
2. Department of Obstetrics and Gynecology, National Yang-Ming University School of Medicine, Taipei, Taiwan;
3. Department of Pharmacy and Master Program, College of Pharmacy and Health Care, Tajen University, Pingtung County, Taiwan;
4. Institute of Biomedical Sciences, National Sun Yat-sen University, Kaohsiung 80424, Taiwan;
5. Department of Emergency Medicine, Taipei Tzu Chi Hospital, Buddhist Tzu Chi Medical Foundation, New Taipei, Taiwan;
6. Department of Emergency Medicine, School of Medicine, Tzu Chi University, Hualien, Taiwan;
7. Department of Biological Science, National Sun Yat-sen University, Kaohsiung, Taiwan;
8. Department of Marine Biotechnology and Resources, National Sun Yat-sen University, Kaohsiung, Taiwan;
9. Marine Biomedical Laboratory and Center for Translational Biopharmaceuticals, Department of Marine Biotechnology and Resources, National Sun Yat-sen University, Kaohsiun, Taiwan;
10. Department of Pathology & Immunology, Baylor College of Medicine, Houston, Texas, USA;
11. School of Medicine, College of Medicine, Fu Jen Catholic University, New Taipei city, Taiwan;
12. Department of Pathology, Show Chwan Memorial Hospital, Changhua, Taiwan
13. Department of Health Food, Chung Chou University of Science and Technology, Changhua, Taiwan
*These authors contributed equally to this work.
Abstract
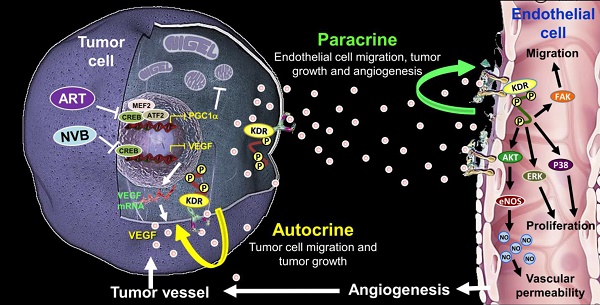
Rationale: Tumor angiogenesis promotes tumor development, progression, growth, and metastasis. Metronomic chemotherapy involves the frequent administration of low-dose chemotherapeutic agents to block angiogenic activity and reduce side effects.
Methods: MDA-MB-231 cells were treated with various concentrations of artemisinin (ART) and vinorelbine (NVB) and the cytotoxic effects of ART/NVB were determined using the CCK-8 assay. Mitochondrial reactive oxygen species (ROS) levels, mitochondrial membrane potential (∆Ψm) and mass were assessed using MitoSOX, TMRE and MitoTracker green staining. Western blot analysis was used to quantify the expression of autophagy-related proteins. Herein, by using bioinformatics analysis and experimental verification, we identified CREB as a master in MDA-MB-231 cells.
Results: We found that artemisinin (ART), which exhibits anti-angiogenic and anti-cancer effects via mitochondrial regulation, synergized with vinorelbine (NVB) to inhibit MDA-MB-231 cell proliferation. ART and NVB cooperated to regulate mitochondrial biogenesis. CREB acted as a crucial regulator of PGC1α and VEGF, which played critical roles in NVB-dependent growth factor depletion. Moreover, CREB suppression significantly reversed mitochondrial dysfunction following ART/NVB co-treatment. In addition, combination treatment with ART and NVB significantly suppressed tumor growth in a nude mouse xenograft model, with downregulated CREB and PGC1α expression levels observed in tumor biopsies, in agreement with our in vitro and ex vivo data.
Conclusions: These findings support the hypothesis that ART affects cancer and endothelial cells by targeting the auto-paracrine effects of VEGF to suppress mitochondrial biogenesis, angiogenesis, and migration between cancer cells and endothelial cells.
Keywords: artemisinin, mitochondria, VEGF, angiogenesis, auto-paracrine
 Global reach, higher impact
Global reach, higher impact