Impact Factor
Theranostics 2021; 11(9):4171-4186. doi:10.7150/thno.42260 This issue Cite
Research Paper
Redox-sensitive carrier-free nanoparticles self-assembled by disulfide-linked paclitaxel-tetramethylpyrazine conjugate for combination cancer chemotherapy
1. Key Laboratory of Coarse Cereal Processing of Ministry of Agriculture and Rural Affairs, Chengdu University, Chengdu 610106, People's Republic of China.
2. State Key Laboratory of Southwestern Chinese Medicine Resources, College of Pharmacy, Chengdu University of Traditional Chinese Medicine, Chengdu 611137, People's Republic of China.
3. School of Basic Medical Sciences, Chengdu University, Chengdu 610106, People's Republic of China.
4. Department of Pharmacology and Pharmacy, University of Hong Kong, Hong Kong 999077, People's Republic of China.
5. College of Materials Science and Engineering, Nanjing Tech Universit`y, Nanjing 211816, People's Republic of China.
Abstract
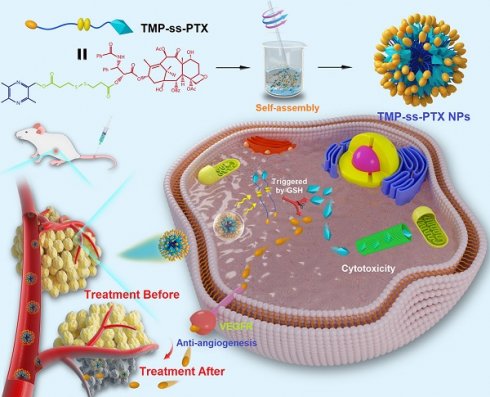
Rationale: Combinations of two or more therapeutic agents targeting different signaling pathways involved in tumor progression can have synergistic anticancer effects. However, combination chemotherapies are greatly limited by the different pharmacokinetics, tumor targeting, and cellular uptake capacities of the combined drugs. We have previously demonstrated the potential synergistic efficacy of paclitaxel (PTX) and the natural anti-angiogenic agent tetramethylpyrazine (TMP) for suppressing ovarian carcinoma growth. An efficient, facile, and smart nanosystem to deliver PTX and TMP simultaneously in vivo is greatly desired.
Methods: We constructed a redox-sensitive nanosystem based on the amphiphilic PTX-ss-TMP conjugate, in which PTX and TMP are linked by a disulfide bond. We characterized the structure of the drug conjugate by 1H NMR and LC-MS, and then prepared PTX-ss-TMP NPs by a one-step nanoprecipitation method. We investigated the redox sensitivity, tumor-targeting ability, anticancer efficacy, and anti-angiogenesis activity of PTX-ss-TMP NPs in vitro and in vivo.
Results: The amphiphilic PTX-ss-TMP conjugate readily self-assembled into stable nanoparticles in aqueous solution with a low critical association concentration of 1.35 µg/mL, well-defined spherical structure, small particle size (152 nm), high drug loading, redox-responsive drug release, high biocompatibility, and high storage stability. In cancer cells pretreated with GSH-OEt, PTX-ss-TMP NPs exhibited higher cytotoxicity, apoptosis rate, and cell-cycle arrest than monotherapy or combination therapy with free drugs, which was attributed to their improved cellular uptake and rapid intracellular drug release. Additionally, PTX-ss-TMP NPs also had a stronger anti-angiogenesis effect in HUVECs than free drug, which was mediated by VEGFR2-involved downstream signals. Finally, PTX-ss-TMP NPs showed tumor-specific accumulation and excellent antitumor activity in A2780 xenograft mice compared with free drug.
Conclusions: These in vitro and in vivo results provide clear evidence that this redox-responsive carrier-free nanosystem with intrinsic amphiphilicity has great potential for combination cancer chemotherapy.
Keywords: drug conjugate, paclitaxel, tetramethylpyrazine, combination chemotherapy, disulfide linkage, self-assembly.
 Global reach, higher impact
Global reach, higher impact