13.3
Impact Factor
Theranostics 2021; 11(9):4187-4206. doi:10.7150/thno.49054 This issue Cite
Research Paper
SARM1 promotes neuroinflammation and inhibits neural regeneration after spinal cord injury through NF-κB signaling
1. Department of Orthopedics (Spine Surgery), The First Affiliated Hospital of Wenzhou Medical University, Wenzhou 325000, Zhejiang, China.
2. School of Basic Medical Sciences, Wenzhou Medical University, Wenzhou 325035, Zhejiang, China.
3. Department of Orthopedics, Taizhou Hospital of Zhejiang Province, Linhai 317000, Zhejiang, China.
4. Engineering Laboratory of Development and Application of Traditional Chinese Medicine from Zhejiang Province and Holistic Integrative Pharmacy Institutes, Hangzhou Normal University, Hangzhou 311121, Zhejiang, China.
5. School of Mental Health, Wenzhou Medical University, Wenzhou 325035, Zhejiang, China.
6. Phase I Clinical Research Center, Zhejiang Provincial People's Hospital of Hangzhou Medical College, Hangzhou, Zhejiang, 310053, China.
*These authors contributed equally to this work.
Abstract
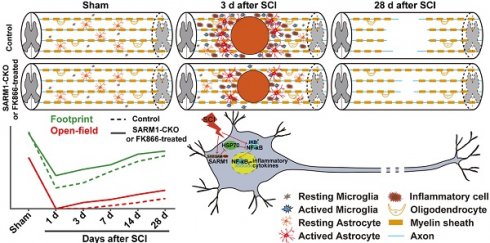
Axonal degeneration is a common pathological feature in many acute and chronic neurological diseases such as spinal cord injury (SCI). SARM1 (sterile alpha and TIR motif-containing 1), the fifth TLR (Toll-like receptor) adaptor, has diverse functions in the immune and nervous systems, and recently has been identified as a key mediator of Wallerian degeneration (WD). However, the detailed functions of SARM1 after SCI still remain unclear.
Methods: Modified Allen's method was used to establish a contusion model of SCI in mice. Furthermore, to address the function of SARM1 after SCI, conditional knockout (CKO) mice in the central nervous system (CNS), SARM1Nestin-CKO mice, and SARM1GFAP-CKO mice were successfully generated by Nestin-Cre and GFAP-Cre transgenic mice crossed with SARM1flox/flox mice, respectively. Immunostaining, Hematoxylin-Eosin (HE) staining, Nissl staining and behavioral test assays such as footprint and Basso Mouse Scale (BMS) scoring were used to examine the roles of SARM1 pathway in SCI based on these conditional knockout mice. Drugs such as FK866, an inhibitor of SARM1, and apoptozole, an inhibitor of heat shock protein 70 (HSP70), were used to further explore the molecular mechanism of SARM1 in neural regeneration after SCI.
Results: We found that SARM1 was upregulated in neurons and astrocytes at early stage after SCI. SARM1Nestin-CKO and SARM1GFAP-CKO mice displayed normal development of the spinal cords and motor function. Interestingly, conditional deletion of SARM1 in neurons and astrocytes promoted the functional recovery of behavior performance after SCI. Mechanistically, conditional deletion of SARM1 in neurons and astrocytes promoted neuronal regeneration at intermediate phase after SCI, and reduced neuroinflammation at SCI early phase through downregulation of NF-κB signaling after SCI, which may be due to upregulation of HSP70. Finally, FK866, an inhibitor of SARM1, reduced the neuroinflammation and promoted the neuronal regeneration after SCI.
Conclusion: Our results indicate that SARM1-mediated prodegenerative pathway and neuroinflammation promotes the pathological progress of SCI and anti-SARM1 therapeutics are viable and promising approaches for preserving neuronal function after SCI.
Keywords: SARM1, spinal cord injury, neuroinflammation, neural regeneration, axonal degeneration
 Global reach, higher impact
Global reach, higher impact