Impact Factor
Theranostics 2021; 11(9):4335-4350. doi:10.7150/thno.52077 This issue Cite
Research Paper
Compensatory combination of mTOR and TrxR inhibitors to cause oxidative stress and regression of tumors
1. The First Affiliated Hospital of Wenzhou Medical University, Wenzhou Medical University, Wenzhou 325035, China.
2. Cancer and Anticancer Drug Research Center, School of Pharmaceutical Sciences, Wenzhou Medical University, Wenzhou 325035, China.
3. Biomedical Collaborative Innovation Center of Zhejiang Province, Wenzhou University, Wenzhou 325035, China.
4. Wenzhou University-Wenzhou Medical University Collaborative Innovation Center of Biomedical, Wenzhou 325035, China.
#These authors contributed equally to this work.
Abstract
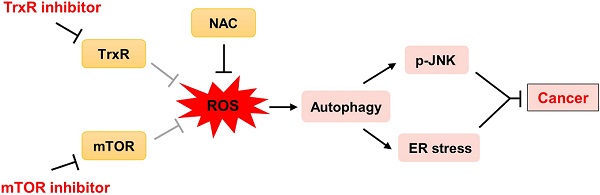
Background: Cancer is a leading cause of death worldwide. Extensive research over decades has led to the development of therapies that inhibit oncogenic signaling pathways. The mammalian target of rapamycin (mTOR) signaling pathway plays an important role in the development of many cancers. Several mTOR inhibitors are approved for the treatment of cancers. However, the anticancer efficacies of mTOR inhibitor monotherapy are still limited.
Methods: Western blot was used to detect the expression of indicated molecules. Thioredoxin reductase (TrxR) activity in cells was determined by the endpoint insulin reduction assay. Immunofluorescence staining was used to analyze precise location and expression of target proteins. Nude mice were used for xenograft tumor models.
Results: We identified a synergistic lethal interaction of mTOR and TrxR inhibitors and elucidated the underlying molecular mechanisms of this synergism. We demonstrated that mTOR and TrxR inhibitors cooperated to induce cell death by triggering oxidative stress, which led to activation of autophagy, endoplasmic reticulum (ER) stress and c-Jun N-terminal Kinase (JNK) signaling pathway in cancer cells. Remarkably, we found that auranofin (AF) combined with everolimus significantly suppressed tumor growth in HCT116 and SGC-7901 xenograft models with no significant signs of toxicity.
Conclusion: Our findings identify a promising therapeutic combination for cancer and has important implications for developing mTOR inhibitor-based combination treatments.
Keywords: mTOR, thioredoxin reductase, oxidative stress, autophagy, c-Jun N-terminal Kinase
 Global reach, higher impact
Global reach, higher impact