13.3
Impact Factor
Theranostics 2021; 11(14):6703-6716. doi:10.7150/thno.57775 This issue Cite
Research Paper
Machine learning-assisted immune profiling stratifies peri-implantitis patients with unique microbial colonization and clinical outcomes
1. Department of Periodontics and Oral Medicine, the University of Michigan School of Dentistry, Ann Arbor, MI 48109.
2. Department of Computational Mathematics, Science, and Engineering, Michigan State University, East Lansing, MI 48823.
3. Department of Biomedical Engineering, College of Engineering & Biointerfaces Institute, University of Michigan, Ann Arbor, MI 48109.
4. Division of Gastroenterology and Hepatology, Department of Internal Medicine, the University of Michigan Medical School, Ann Arbor, MI 48105.
5. Rogel Cancer Center, the University of Michigan, Ann Arbor, MI 48105.
6. Current Affiliation: Department of Oral Medicine, Infection, and Immunity, Harvard School of Dental Medicine, Boston, MA 02115.
Abstract
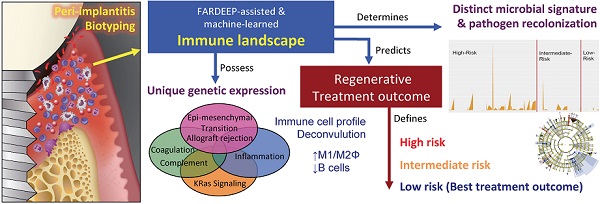
Rationale: The endemic of peri-implantitis affects over 25% of dental implants. Current treatment depends on empirical patient and site-based stratifications and lacks a consistent risk grading system.
Methods: We investigated a unique cohort of peri-implantitis patients undergoing regenerative therapy with comprehensive clinical, immune, and microbial profiling. We utilized a robust outlier-resistant machine learning algorithm for immune deconvolution.
Results: Unsupervised clustering identified risk groups with distinct immune profiles, microbial colonization dynamics, and regenerative outcomes. Low-risk patients exhibited elevated M1/M2-like macrophage ratios and lower B-cell infiltration. The low-risk immune profile was characterized by enhanced complement signaling and higher levels of Th1 and Th17 cytokines. Fusobacterium nucleatum and Prevotella intermedia were significantly enriched in high-risk individuals. Although surgery reduced microbial burden at the peri-implant interface in all groups, only low-risk individuals exhibited suppression of keystone pathogen re-colonization.
Conclusion: Peri-implant immune microenvironment shapes microbial composition and the course of regeneration. Immune signatures show untapped potential in improving the risk-grading for peri-implantitis.
Keywords: peri-implantitis, classification, immune profiling, microbiome, FARDEEP
 Global reach, higher impact
Global reach, higher impact