Impact Factor
Theranostics 2022; 12(3):1132-1147. doi:10.7150/thno.66325 This issue Cite
Research Paper
Bioorthogonal catalytic nanozyme-mediated lysosomal membrane leakage for targeted drug delivery
1. College of Pharmacy, and State Key Laboratory of Medicinal Chemical Biology, Nankai University, Tianjin 300350, China.
2. Key Laboratory of Bioactive Materials for the Ministry of Education, College of Life Sciences, Nankai University, Tianjin 300071, China.
3. School of Medicine, Nankai University, Tianjin 300071, China.
4. School of Materials Science and Engineering, Nankai University, Tianjin 300350, China.
5. State Key Laboratory of Biocatalysis and Enzyme Engineering, Hubei Key Laboratory of Industrial Biotechnology, School of Life Sciences, Hubei University, Wuhan 430062, China.
6. Joint Laboratory of Nanozymes, College of Life Sciences, Nankai University, Tianjin 300071, China.
7. CAS Engineering Laboratory for Nanozymes, Institute of Biophysics, Chinese Academy of Sciences, Beijing 100101, China.
Abstract
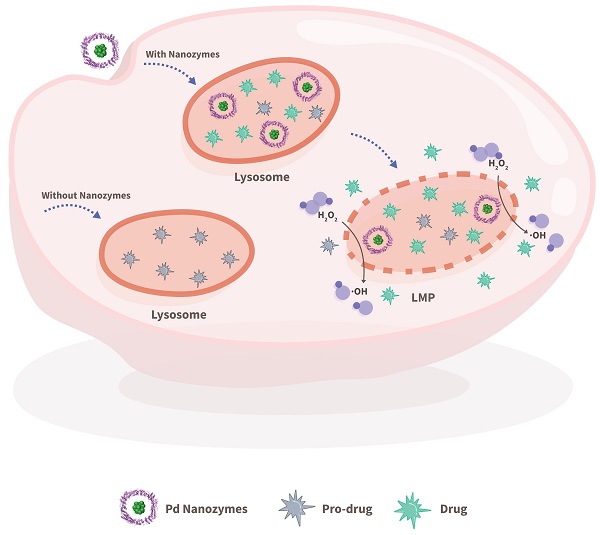
Rationale: Employing in situ bioorthogonal catalysis within subcellular organelles, such as lysosomes, remains a challenge. Lysosomal membranes pose an intracellular barrier for drug sequestration, thereby greatly limiting drug accumulation and concentrations at intended targets. Here, we provide a proof-of-concept report of a nanozyme-based strategy that mediates in situ bioorthogonal uncaging reactions within lysosomes, followed by lysosomal escape and the release of uncaged drugs into the cytoplasm.
Methods: A model system composed of a protein-based nanozyme platform (based on the transition metals Co, Fe, Mn, Rh, Ir, Pt, Au, Ru and Pd) and caged compound fluorophores was designed to screen for nanozyme/protecting group pairings. The optimized nanozyme/protecting group pairing was then selected for utilization in the design of anti-cancer pro-drugs and drug delivery systems.
Results: Our screening system identified Pd nanozymes that mimic mutant P450BM3 activity and specifically cleave propargylic ether groups. We found that the intrinsic peroxidase-like activity of Pd nanozymes induced the production of free radicals under acid conditions, resulting in lysosomal membrane leakage of uncaged molecules into the cytoplasm. Using a multienzyme synergistic approach, our Pd nanozymes achieved in situ bioorthogonal catalysis and nanozyme-mediated lysosomal membrane leakage, which were successfully applied to the design of model pro-drugs for anti-cancer therapy. The extension of our nanozyme system to the construction of a liposome-based “all-in-one” delivery system offers promise for realizing efficacious in vivo tumor-targeted therapies.
Conclusions: This strategy shows a promising new direction by utilizing nanotechnology for drug development through in situ catalyzing bioorthogonal chemistry within specific subcellular organelles.
Keywords: Nanozyme, Lysosome, Bioorthogonal catalysis, Pro-drug, Targeted delivery, Tumor
 Global reach, higher impact
Global reach, higher impact