13.3
Impact Factor
Theranostics 2022; 12(10):4548-4563. doi:10.7150/thno.71378 This issue Cite
Research Paper
NOTCH1 signaling regulates the latent neurogenic program in adult reactive astrocytes after spinal cord injury
Institute of Neuroscience, Key Laboratory of Molecular Neurobiology of Ministry of Education and the Collaborative Innovation Center for Brain Science, Naval Medical University, Shanghai 200433, China.
* These authors contributed equally to this work
Abstract
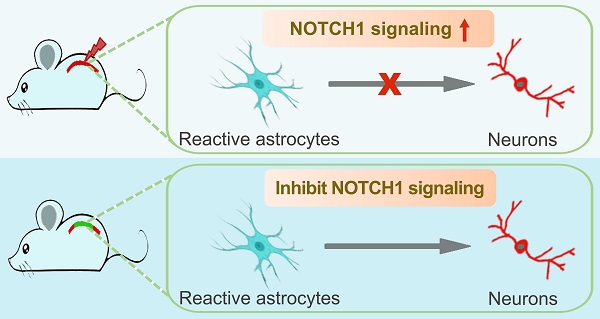
Background: Direct reprogramming of astrocytes into neurons opens up a new avenue for neuroregenerative medicine. However, the poor understanding of the molecular mechanisms underpinning the latent neurogenic program in astrocytes has largely restricted this strategy towards safe and effective clinical therapies.
Methods: Immunocytochemistry, immunohistochemistry, western blotting, qRT-PCR, gene knockdown and fate-mapping are performed to analyze the role of NOTCH1 signaling in regulation of the latent neurogenic program in reactive astrocytes after spinal cord injury.
Results: Western blotting analysis highlights that NOTCH1 is a key signaling mediating Ascl1- and Neurog2-driven astrocyte-to-neuron conversion. Inhibition of NOTCH1 signaling in cultured astrocytes by shRNA or DAPT (a NOTCH1 inhibitor) is sufficient to reprogram them into neurons by upregulating the expression of pro-neural transcription factors, including NeuroD1, NeuroD2, Pax6, Lmx1a and Lhx6. In the spinal cord of adult mouse, the expression of Notch1 is detected in resident astrocytes, which was significantly increased after spinal cord injury (SCI). Genetical knockdown of NOTCH1 signaling alone successfully triggers endogenous reactive astrocytes reprogramming into neurons in the injured adult spinal cord. Importantly, pharmacologically blocking NOTCH1 signaling with small molecule DAPT alone can also induce in situ astrocyte-to-neuron conversion after SCI.
Conclusions: We identify NOTCH1 as a key common signaling pathway in reactive astrocyte that provides a barrier for cell fate conversion. This proof-of-principle study will significantly expand our molecular understanding of astroglial-lineage reprogramming and overcoming the NOTCH1 gatekeeper with small molecules may provide a transgene-free approach for in vivo chemical neuronal reprogramming with potential clinical application in neuroregeneration.
Keywords: NOTCH1, small molecule, astrocyte-to-neuron conversion, in vivo reprogramming, spinal cord injury
 Global reach, higher impact
Global reach, higher impact