13.3
Impact Factor
Theranostics 2022; 12(13):5856-5876. doi:10.7150/thno.73386 This issue Cite
Research Paper
Nanogel-mediated delivery of oncomodulin secreted from regeneration-associated macrophages promotes sensory axon regeneration in the spinal cord
1. Department of Brain Science, Ajou University Graduate School of Medicine, Suwon, 16499, Republic of Korea.
2. Neuroscience Graduate Program, Department of Biomedical Sciences, Ajou University Graduate School of Medicine, Suwon, 16499, Republic of Korea.
3. Department of Pharmacy, College of Pharmacy, The Catholic University of Korea, Bucheon, 14662, Republic of Korea.
4. Department of Microbiology, Ajou University School of Medicine, Suwon 16499, Republic of Korea.
5. Department of Pharmacology, Ajou University School of Medicine, Suwon, 16499, Republic of Korea.
6. Center for Convergence Research of Neurological Disorders, Ajou University School of Medicine, Suwon, 16499, Republic of Korea.
7. Department of Neurology, Ajou University School of Medicine, Suwon, 16499, Republic of Korea.
8. AI-Superconvergence KIURI Translational Research Center, Suwon, 16499, Republic of Korea.
Abstract
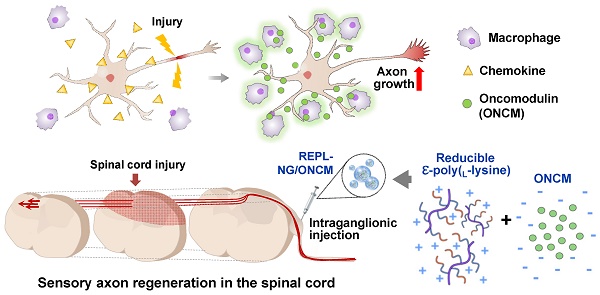
Preconditioning nerve injury enhances axonal regeneration of dorsal root ganglia (DRG) neurons in part by driving pro-regenerative perineuronal macrophage activation. How these macrophages influence the neuronal capacity of axon regeneration remains elusive. We report that oncomodulin (ONCM) is produced from the regeneration-associated macrophages and strongly influences regeneration of DRG sensory axons. We also attempted to promote sensory axon regeneration by nanogel-mediated delivery of ONCM to DRGs.
Methods: In vitro neuron-macrophage interaction model and preconditioning sciatic nerve injury were used to verify the necessity of ONCM in preconditioning injury-induced neurite outgrowth. We developed a nanogel-mediated delivery system in which electrostatic encapsulation of ONCM by a reducible epsilon-poly(L-lysine)-nanogel (REPL-NG) enabled a controlled release of ONCM.
Results: Sciatic nerve injury upregulated ONCM in DRG macrophages. ONCM in macrophages was necessary to produce pro-regenerative macrophages in the in vitro model of neuron-macrophage interaction and played an essential role in preconditioning-induced neurite outgrowth. ONCM increased neurite outgrowth in cultured DRG neurons by activating a distinct gene set, particularly neuropeptide-related genes. Increasing extracellularly secreted ONCM in DRGs sufficiently enhanced the capacity of neurite outgrowth. Intraganglionic injection of REPL-NG/ONCM complex allowed sustained ONCM activity in DRG tissue and achieved a remarkable long-range regeneration of dorsal column sensory axons beyond spinal cord lesion.
Conclusion: NG-mediated ONCM delivery could be exploited as a therapeutic strategy for promoting sensory axon regeneration following spinal cord injury.
Keywords: Oncomodulin, Axon regeneration, Nanogel, Spinal cord injury, Regeneration-associated macrophages
 Global reach, higher impact
Global reach, higher impact