Impact Factor
Theranostics 2023; 13(12):4266-4286. doi:10.7150/thno.84971 This issue Cite
Review
Engineering strategies and optimized delivery of exosomes for theranostic application in nerve tissue
1. Department of Trauma and Orthopedics, Peking University People's Hospital, Beijing 100000, China.
2. Key Laboratory of Trauma and Neural Regeneration (Peking University), Ministry of Education, Beijing 100000, China.
3. National Center for Trauma Medicine, Beijing 100000, China.
4. Department of Central Laboratory and Institute of Clinical Molecular Biology, Peking University People's Hospital, Beijing 100000, China.
Received 2023-4-5; Accepted 2023-7-5; Published 2023-7-24
Abstract
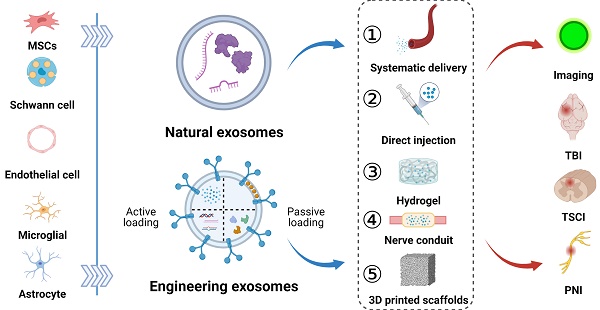
Severe injuries or diseases affecting the peripheral and central nervous systems can result in impaired organ function and permanent paralysis. Conventional interventions, such as drug administration and cell-based therapy, exhibit limited effectiveness due to their inability to preserve post-implantation cell survival and impede the deterioration of adjacent tissues. Exosomes have recently emerged as powerful tools for tissue repair owing to their proteins and nucleic acids, as well as their unique phospholipid properties, which facilitate targeted delivery to recipient cells. Engineering exosomes, obtained by manipulating the parental cells or directly functionalizing exosomes, play critical roles in enhancing regenerative repair, reducing inflammation, and maintaining physiological homeostasis. Furthermore, exosomes have been shown to restore neurological function when used in combination with biomaterials. This paper primarily focuses on the engineering strategies and delivery routes of exosomes related to neural research and emphasizes the theranostic application of optimized exosomes in peripheral nerve, traumatic spinal cord, and brain injuries. Finally, the prospects of exosomes development and their combination with other approaches will be discussed to enhance our knowledge on their theranostic effectiveness in neurological diseases.
Keywords: biomaterials, engineering exosomes, exosomal miRNA, nerve injury, neuroimaging
Introduction
The central and peripheral nervous systems (CNS and PNS, respectively) are the most complex conduction systems in the human body. Severe injuries or diseases pose challenges to the recovery of the peripheral nerve, spinal cord, and brain because of substantial defects in the axons, vasculature, and neuronal deficits [1, 2]. Conventional treatments involve the application of drugs or crosslinked polymers, including natural biomaterials (e.g., chitosan, collagen, and fibrin) or synthetic materials (e.g., PLGA and PLLA), which have been developed to facilitate the loading of the neural stem cells (NSCs), mesenchymal stem cells (MSCs), and growth factors for neural tissue repair [3, 4]. However, the following challenges still persist in implementing these therapeutic strategies: (1) administration of high-dose hormones can result in serious side effects, and the presence of biological barriers prevents traditional drugs from reaching injured sites [5]; (2) living cells loaded into biomaterials may not receive adequate blood supply during early transplantation, leading to decreased cell viability or cell death; (3) injectable biomaterials loaded with NSCs or MSCs require extensive clinical trials to prove their safety and efficacy in the human body; and (4) the short half-life and inadequate intracellular delivery of growth factors restrict their potential to promote the long-term growth of neighboring cells and axons.
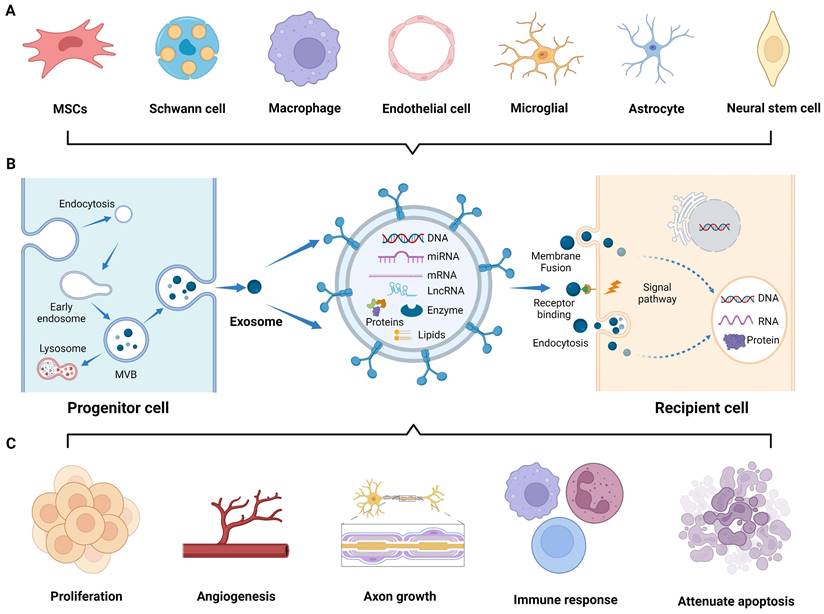
To overcome these limitations, researchers have devoted considerable effort to studying extracellular vesicles (EVs), including microvesicles, apoptotic bodies, and exosomes [6]. In particular, exosomes, which can be biosynthesized and secreted by various cells (Figure 1A) [7-9], possess a nano-scale membrane structure (30-100 nm) that enables them to cross biological barriers. Moreover, upon entering the circulation, exosomes exhibit resistance to rapid degradation, and their specific molecules on the lipid bilayer shell allow them to target and fuse with distant cell membranes, thereby mediating intercellular communication. Additionally, the internal cavity of exosomes encapsulates a large number of signaling moieties, such as enzymes, transcription factors, and cytokines. Furthermore, the bilayer membrane structure exhibits hydrophilic and hydrophobic properties, endowing exosomes with cell-like carrier characteristics for tissue engineering purposes [10, 11]. In the field of regenerative medicine, the utilization of biomaterials as exosomes carriers can achieve sustained and localized release of exosomes, which enhances the reparative effects mediated by biomaterials that are not loaded with exosomes [12]. Extensive studies have been conducted on natural and engineered exosomes in animal models of neurological injuries, such as sciatic nerve, traumatic spinal cord, and brain injuries. In these studies, many similarities were observed in the therapeutic principles and delivery pathways, owing to the close structural relationship between the PNS and CNS.
Application of exosomes in nerve regeneration. (A) Exosomes derived from various cells research. (B) Biogenesis and interaction of exosomes with recipient cells. (C) Application of exosomes to the sites of nerve injury, promoting cell proliferation, angiogenesis, and axon growth, modulating immune response, and attenuating cell apoptosis. Figures created with Biorender.com.
Currently, there have been numerous reports on the preparation, transport mechanisms, and application of exosomes; however, existing literature lacks comprehensive reviews on the engineering strategies of exosomes and their application in nerve tissues. In this review, we will highlight the methods of functionalizing exosomes at the cellular and exosomal levels to enhance their efficacy in neuroregeneration and their delivery routes. We also discuss the application of exosomes in neuroimaging and their combination with nerve conduits, hydrogels, and 3D printed bioscaffold for treating peripheral nerve, traumatic spinal cord, and brain injuries, providing new insights into future developments in the field of regenerative medicine for nerve tissues.
Biogenesis, interaction, and tissue source
In 1987, Johnstone et al. first described extracellular “exosomes” as a mechanism for shedding of specific membrane functions [13]. Since then, the biogenesis of exosomes has been continuously reported, and it is now described as an endocytic pathway originating from cell membrane. Briefly, exosomes generation is usually divided into four steps: invagination, endosome formation, fusion, and secretion [14]. The invagination of the cell membrane forms clathrin-coated vesicles by inward budding and then fuses together to form early endosomes in the cytoplasm. In the maturation process of endosomes, intraluminal vesicles (ILVs) begin to form through further invagination of the limiting endosomal membrane, a crucial step regulated by the endosomal sorting complex required for transport (ESCRT). Subsequently, the ILVs containing proteins, lipids, and nucleic acids develop into multivesicular bodies (MVBs). Some MVBs are transported to the lysosomes for degradation, while others fuse with the plasma membrane and release exosomes into the extracellular microenvironment (Figure 1B) [15]. The released exosomes primarily interact with recipient cells through three routes [16]: (1) complete endocytosis by the recipient cells, (2) direct membrane fusion between exosomes and recipient cells, and (3) direct interaction of transmembrane proteins on the exosomes with signaling molecules on the surface of the recipient cells, which play an important role in mediating gene expression and signaling pathways of the recipient cells by transferring the contents and biological signals carried by the exosomes (Figure 1B) [17].
In the field of neuroprotection research, numerous studies have demonstrated the role of exosomes originating from cells of the CNS, PNS, mesenchymal tissues, and other tissues in nerve regeneration. Exosomes derived from the CNS, including the brain and spinal cord, are released by the neurons [17], astrocytes [18], microglia [19], and oligodendrocytes [20]. These exosomes specifically target brain and spinal cord diseases. The PNS is another valuable source of tissue-derived exosomes that have regenerative potential. Exosomes derived from the Schwann cells (SCs), the principal glial cells in the PNS, have been extensively studied [21]. Furthermore, exosomes derived from other PNS components, such as macrophages, contribute to neural repair processes [22]. Moreover, exosomes derived from various mesenchymal tissues, such as the bone marrow, adipose tissue, and umbilical cord, have shown promise in promoting nerve regeneration [23]. Exosomes released by MSCs within these tissues carry diverse cargo molecules, including growth factors, cytokines, and membrane proteins, thereby promoting PNS and CNS repair and avoiding the risks associated with MSCs transplantation. Besides, exosomes derived from other tissues, such as the blood vessel and muscle, exert regenerative effects on injured nerves [8, 24]. These tissue-derived exosomes enter the injured nerve area and participate in various regulatory events, including promoting axonal myelinization and angiogenesis; regulating immune responses; reducing cell apoptosis; and facilitating peripheral nerve, spinal cord, and brain injury repair (Figure 1C) [25-27]. Nonetheless, the biofunction and yield of natural exosomes are inherently limited, underscoring the necessity to engineer both source cells and exosomes to enhance their theranostic potential in the nervous system.
Pretreatment of progenitor cells to obtain engineering exosomes
Studies have shown that cell sources can regulate the contents of exosomes. For instance, the tissue source of MSCs determines the heterogeneity of proteins within secreted exosomes, leading to functional differences, as revealed by bioinformatics analysis [28]. Furthermore, parental cells can promptly respond to environmental conditions and modulate the yield and bioactivity of nanovesicles, which is fundamental for achieving large-scale production and maintaining homeostasis. Therefore, various approaches at the cellular level have been employed to promote exosome production, and genetic engineering techniques can also be used to specialize them for enhancing tissue regeneration (Figure 2).
Multiple factors, including culture conditions, biochemical factors, mechanical stimulation, and genetic engineering, can affect the yield and biofunctions of exosomes derived from source cells. Figures created with Biorender.com.
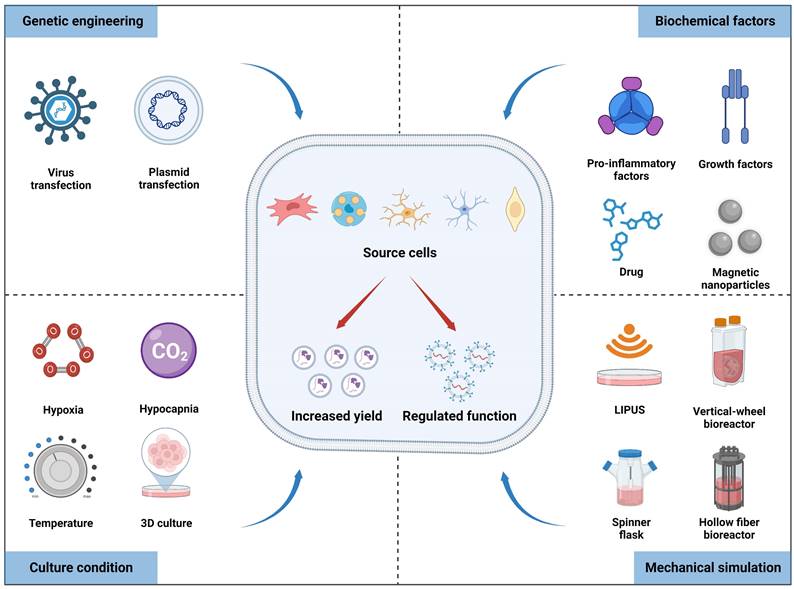
Cell culture conditions
Cellular functions and phenotypes can be altered under different culture conditions, indirectly affecting the production and bioactivity of exosomes obtained from pretreated cells (Table 1). Previous studies have revealed that stem cells cultured under high-density conditions exhibit contact inhibition and enter a quiescent state [29]. Conversely, lower-density cultivation can activate paracrine signaling pathways and promote exosomes secretion; therefore it is preferred to collect the supernatant for exosomes separation when cell confluence reaches 60% to 90% [30]. Hypoxic preconditioning is a frequently used strategy to improve therapeutic outcomes prior to stem cell transplantation. Prior investigations have revealed that hypoxic preconditioning affects gene expression and proteome of the dental pulp stem cells cultured in a 3D system compared to normoxic conditions, resulting in the upregulation of the HIF-1α, VEGFA, and KDR mRNA, along with proteins involved in angiogenesis [31]. Interestingly, exosomes produced from hypoxia-preconditioned human umbilical cord MSCs (hUCMSCs) group exhibited increased HIF-1α content compared to that of the normoxic group, thereby enhancing the pro-angiogenic effects in spinal cord injury via VEGF overexpression in the surrounding transplantation area [32]. Consequently, hypoxic preconditioning may be an effective and promising method for exosomes engineering. Additionally, cells cultured in 3D culture system exhibit closer resemblance to in vivo state compared to traditional 2D culture methods, thereby augmenting the paracrine effects of MSCs and optimizing exosomes function [33]. For example, UCMSCs cultured in a microcarrier-based 3D system resulted in a 20-fold increase in exosomes production compared to that of 2D culture. Additionally, when combined with tangential flow filtration (TFF), exosomes production was further amplified by seven times compared to 3D-derived exosomes alone. The delivery efficiency of siRNA to neurons using 3D-TFF-exosomes also increased by seven times [34]. In conclusion, integrating diverse culture conditions with advanced isolation techniques can substantially boost the scale-up production and activity of exosomes.
List of multiple factors to enhance exosomes bioactivity
| Extracellular factors | Exosomes source | Effect on exosomes | Molecular mechanism | References | |
|---|---|---|---|---|---|
| Culture conditions | Low culture density | BMSCs | Increased production of exosomes | N/A | [30] |
| Hypoxia | UCMSCs | Angiogenesis, promoted functional recovery after injury | HIF-1α↑, VEGF↑ | [32] | |
| 3D culture | UCMSCs | Increased production of exosomes and siRNA delivery to neurons | N/A | [34] | |
| Mild hypothermia | Microglia | Upregulated the expression of miR-20b-5p, promoted neurite outgrowth and synapse recovery | PTEN↓, PI3K/AKT↑ | [47] | |
| Hypocapnia | ADMSCs | Promoted cellular activity and regeneration of nerve fibers | N/A | [48] | |
| Biochemical factors | LPS, IFN-γ, TNF-α | BMSCs | Anti-inflammation | NF-κB↓, AKT1/AKT2↑; COX2/PGE2↑ | [35, 36] |
| Fe3O4, Iron oxide nanoparticles | BMSCs | Angiogenesis, targeting injured sites | SPRY2↓, PI3K/AKT and ERK1/2↑; N/A | [37, 38] | |
| IGF-1 | NSCs | Enrichment of miRNA level, anti-inflammation, anti-apoptosis | miR-219a-2-3p↑, YY1↓, NF-κB↓ | [49] | |
| BDNF | UCMSCs, BMSCs | Remodeled neural networks, inhibited apoptosis | N/A; miR-216a-5p↑ | [50, 51] | |
| FK506 | ADMSCs | Enhanced nerve regeneration, reduced autophagy, enrichment of protein level | N/A; HSPA8 and EEF1A1↑ | [52, 53] | |
| Mechanical factors | Hollow fiber bioreactors | UCMSCs | Angiogenesis | TGF-β1 and Smad2/3↑ | [44] |
| Vertical-wheel bioreactors | BMSCs, ADMSCs, UCMSCs | Increased production of exosomes | N/A | [45] | |
| Low-intensity pulsed ultrasound (LIPUS) | Schwann cells | Enhanced axon elongation, promoted SCs proliferation | PI3K/AKT/FoxO↑ | [46] | |
| Environmental enrichment | Youthful systemic milieu | Serum | Promoted OPC differentiation and myelination | miR-219↑ | [54] |
| Physical, intellectual and social activities | Immune cells | Promoted CNS myelination, reduced LPS-induced astrogliosis | miR-219↑ | [55] | |
Generation of engineering exosomes via biochemical factors stimulation
The production and function of exosomes can be regulated by pretreating parental cells with biochemical factors, such as pro-inflammatory factors, magnetic nanoparticles, and growth factors (Table 1). Xu et al. treated MSCs with lipopolysaccharide (LPS), a typical pro-inflammatory factor [35]. The generated exosomes significantly reduced the secretion of pro-inflammatory factors from LPS-stimulated RAW 264.7 cells, without affecting cell viability and apoptosis. The results of the western blot assay showed that AKT1 and AKT2 phosphorylation was activated through the NF-κB signaling pathway in M2 and M1 macrophages, which downregulated TNF-α, IL-6, and IL-1, thereby reducing inflammation response.
Harting et al. induced the secretion of anti-inflammatory exosomes from the bone mesenchymal stem cells (BMSCs) under IFN-γ and TNF-α stimulation to precisely target inflammation sites [36]. Although the size distribution and surface markers of exosomes derived from inflammation-treated and naive MSCs were similar, differences were observed in their protein composition, cytokines, and RNA content, with further attenuation of the release of pro-inflammatory cytokines, partly due to the expression of COX2/PEG2 compared to naive MSCs-derived exosomes. The combined effects of exosomes and cytokines on immune regulation provide new insights into intercellular communication research. In addition to pro-inflammatory factors, Fe3O4 and iron oxide nanoparticles have been proven to regulate BMSCs-derived exosomes, exhibiting targeting and pro-angiogenic potential [37, 38]. The engineered exosomes contained high levels of miR-21-5p, which promoted the expression of VEGF and HIF-1α in HUVECs by inhibiting SPRY2 and activating the PI3K/AKT and ERK1/2 signaling pathways, leading to cell proliferation and tubulogenesis. However, exosomes derived from high glucose-treated SCs further exacerbated the sciatic nerve impairment in diabetic db/db mice [39]. In summary, progenitor cells preconditioned with appropriate biochemical factors can effectively improve the functional properties of exosomes and facilitate nerve repair.
Improvement of exosomes yield via mechanical factors
Studies have found that mechanical stimulation, such as matrix stiffness, compressive, tensile, and shear stress, can modulate cellular characteristics [40, 41]. For instance, an increase in fluid shear stresses promoted osteogenic differentiation and adipogenic dedifferentiation of MSCs via YAP expression [42]. In recent years, many studies have displayed that mechanical cues alter the yield and biofunction of nanovesicles produced by progenitor cells. The shear stresses generated from dynamic systems, such as shake flasks, spinner flasks, roller bottles or bioreactors (mixing bubbles from spargers) affect cell phenotype and exosomes production [43]. Yan et al. used a hollow-fiber bioreactor to culture UCMSCs and obtained exosomes with favorable biological functions and high yield (Table 1) [44]. The hollow-fiber bioreactor possesses a large surface area that enables high-density cell seeding and facilitates the delivery of nutrients and oxygen to cells, while simultaneously diffusing and expelling cell metabolic waste through the hollow fibers. Compared to traditional 2D culture flasks, the bioreactor yielded 7.5-fold increase in the quantity of exosomes. Additionally, the 3D-derived exosomes exhibited significant advantages in promoting proliferation, migration, and matrix synthesis of recipient cells, which may be related to the activation of TGF-β1 and Smad2/3 signaling pathways. Similarly, Fuzeda et al. utilized a vertical-wheel bioreactor to culture MSCs from the bone marrow, adipose tissue, and umbilical cord seeded onto microcarriers (Table 1) [45]. The shear stress exerted on cell membrane led to an increase in surface tensile strength and membrane contraction. Ultimately, the concentration and productivity of exosomes increased by 5.7-fold and 3-fold, respectively, compared with the static system. Apart from the aforementioned bioreactors, ultrasonic waves can generate shear stress on cell membranes. Ye et al. isolated exosomes derived from low-intensity pulsed ultrasound (LIPUS)-stimulated SCs to treat cavernous nerve crush injury [46]. The results showed that LIPUS stimulation altered the miRNA expression of SCs-exosomes and enhanced nerve regeneration through the PI3K-Akt-FoxO signaling pathway (Table 1). These findings suggested that a scalable culture of exosomes-originating cells using mechanical stimulation can produce high-yield and functionally enhanced nanovesicles for the development of novel therapeutic products.
Specific gene transfected into progenitor cells to alter exosomal content
Exosomes carry a diverse array of contents from donor cells and possess high heterogeneity, playing an important role in regulating the function of recipient cells. In 2007, Valadi et al. first discovered that mast cell-derived exosomes carried mRNA and miRNA, and the transferred exosomal mRNA can be translated into functional proteins in recipient cells [56]. In addition, nucleic acid cargos loaded into exosomes also include DNA, ribosomal RNA (rRNA), long non-coding RNA (lncRNA), transfer RNA (tRNA), small nuclear RNA (snRNA), small nucleolar RNA, and p-element-induced wimpy testis (piwi)-interacting RNA [19]. Among these, microRNA, the most abundant RNA species in exosomes, is a type of non-coding single-stranded RNA molecule encoded by endogenous genes and approximately 17-24 nucleotides in length; it participates in various biological processes in exosomes-mediated intercellular communication, such as angiogenesis, cell transport, cell apoptosis, and protein degradation [7, 16, 57].
Recent scientific reports on the utilization of miRNAs as gene carriers in exosomes for the diagnosis and treatment of neurological diseases have increased in number [58, 59]. To enhance the regeneration of damaged axons, researchers have used genetic engineering techniques to deliver specific miRNAs, carried by viruses or plasmids, into recipient cells such as the MSCs, Schwann cells, and microglia, thereby increasing the production of engineered exosomes. Some studies have shown that MSCs overexpressing miRNAs are more effective at promoting functional recovery than normal MSCs. The overexpression of miR-133b in MSCs has been shown to improve the recovery of hindlimb function and promote axon regeneration in rats following compression spinal cord injury, achieved through the modulation of exosomal miR-133b, which activated ERK1/2, STATS, and CREB signaling pathway proteins [60]. Exosomes and their miRNA cargos derived from MSCs also have immunoregulatory properties. Exosomes containing miR-125a derived from BMSCs reduced inflammatory responses and promoted M2 polarization. This study revealed the neuroprotective role of exosomal miR-125a through the negative regulation of IRF5 expression in SCI rats [61]. Moreover, miRNA-loaded exosomes from different cells promote nerve repair by diminishing cell apoptosis, autophagy, and pyroptosis. Liang et al. discovered that the exosomal miR-499a-5p contributed to reducing neuronal apoptosis after OGD/R and confirmed its inhibitory effect on the JNK3/c-jun signaling pathway by targeting JNK3 [26]. Exosomes derived from ADMSCs, transported miRNA-26b to injured Schwann cells, leading to the suppression of cell autophagy, which was related to the downregulation of Kpna2. On the other hand, M2 microglia-exosomes enriched with miR-672-5p suppressed the AIM2/ASC/caspase-1 signaling pathway via inhibiting AIM2 activity, thereby negatively regulating neuronal pyroptosis and ultimately fostering the functional recovery of the spinal cord. Overall, increasing evidence of the involvement of exosomes and miRNAs in the treatment of nerve injuries highlights the potential of these molecules as therapeutic targets. The molecular mechanism of miRNA-containing exosomes in nerve repair is listed in Table 2.
Molecular mechanism of miRNA-containing exosomes in nerve repair
| microRNA | Cell source | Biological function | Molecular mechanism | Application | References |
|---|---|---|---|---|---|
| miR-22-3p | ADMSCs | Enhanced SCs proliferation and migration | PTEN↓, p-AKT/mTOR↑ | Peripheral nerve injury | [62] |
| miR-26b | ADMSCs | Promoted myelination, reduced autophagy of injured SCs | Kpna2↓ | Sciatic nerve crush injury | [63] |
| miR-214 | MDSCs | Improved SCs function and axon extension | PTEN↓, JAK2/STAT3↑ | Sciatic nerve crush injury | [24] |
| miR-199a-3p/145-5p | UCMSCs | Promoted PC12 cell differentiation, facilitated spinal cord functional recovery | NGF/TrkA↑ | Traumatic spinal cord injury | [64] |
| miR-672-5p | M2 microglial | Promoted axon regeneration, reduced neural pyroptosis | AIM2/ASC/caspase-1↓ | Traumatic spinal cord injury | [65] |
| miR-499a-5p | ADMSCs | Reduced neuronal apoptosis, improved the functional recovery | JNK3/c-jun↓ | Spinal cord injury | [26] |
| miR-133b | ADMSCs | Promoted expressions of NF, GAP-43, GFAP, and MBP | p-CREB/CREB↑, p-STAT3/STAT3↑ | Spinal cord injury | [66] |
| miR-125a | BMSCs | Promoted the recovery of locomotor function and M2 polarization, inhibited neuronal apoptosis and inflammatory response | IRF5↓ | Spinal cord injury | [61] |
| miR-133b | BMSCs | Preserved neurons, promoted axon regeneration, improved the recovery of locomotor function | ERK1/2, STAT3, CREB↑ | Spinal cord injury | [60] |
| miR-124-3p | BMSCs | Impeded cell apoptosis, enhanced M2 polarization | Ern1↓ | Spinal cord ischemia-reperfusion injury | [67] |
| miR-124-3p | microglia | Promoted M2 microglial polarization, inhibited neuronal autophagy, | PDE4B/mTOR↓; FIP200↓ | Traumatic brain injury | [19, 68] |
| miR-21-5p | HT-22 neurons | Inhibited neuronal autophagy | Rab11a↓ | Traumatic brain injury | [69] |
| miR-873a-5p | Astrocytes | Attenuated microglia-mediated neuroinflammation, improved neurological deficits | NF-κB↓ | Traumatic brain injury | [18] |
| miR-181b | BMSCs | Reduced apoptosis and neuroinflammation, promoted M2 microglia polarization | IL10/STAT3↑ | Traumatic brain injury | [70] |
| miR-182-5p | Astrocytes | Inhibited neuroinflammation | Rac1↓ | MCAO/R | [71] |
| miR-17-92 cluster | BMSCs | Promoted axonal growth, enhanced neuroplasticity and functional recovery | PTEN/mTOR↑; PI3K/AKT/mTOR/GSK-3β↑ | Brain injury (traumatic brain injury, MCAO) | [72, 73] |
Direct functionalization of exosomes for nervous system
Although exosomes can be engineered at the cellular level to enhance their yield and function through the mechanism of protein production from progenitor cells, specific engineering tools, such as fluorescent probes and siRNA, cannot be naturally synthesized in parental cells [74]. Cell engineering strategies limit the clinical potential of natural exosomes for in vivo imaging and gene therapy. Consequently, exosomes should be designed within the extracellular environment to integrate the diagnosis and treatment of nanoscale vesicles based on the methods of incubation with cargos and membrane permeabilizer, extrusion, electroporation, sonication, freeze and thaw cycles, liposome-based transfection, and click chemistry [75].
These methods for the direct functionalization of exosomes are as follows (Figure 3): (1) Incubation with exosomes involves the binding of hydrophobic cargo to the bilipid layer of the exosomes. Typically, macrophage-derived exosomes were co-incubated with the hydrophobic drug curcumin to produce curcumin-containing exosomes that targeted the cerebral ischemia-reperfusion region [76]. (2) Surfactant saponin is usually used as membrane permeabilizer to form a complex with cholesterol on the exosomes membrane to increase the incorporation of external drugs. Exosomes loaded with catalase using saponin permeabilization significantly increased the neuroprotective effects compared with those of the control group, whereas incubation at room temperature without saponin showed no difference [77]. (3) To integrate the exosomes membrane, serial extrusion using a lipid extruder is required. Khongkow et al. mixed exosomes and gold nanoparticles (GNPs) in PBS and used a mini extruder to serially extrude them through 400 nm, 200 nm, and 100 nm polycarbonate porous membranes, obtaining brain-targeted exosomes coated with GNPs [78]. (4) Electroporation is another method that uses high voltage generated by an electric field to create temporary pores in the exosomes membrane, which is particularly advantageous for siRNA loading. Huang et al. used electroporation to load CTGF gene inhibitors into MSCs-derived exosomes to attenuate spinal cord injury [79]. (5) Sonication-produced mechanical shear forces result in the loss of exosomes membrane integrity. The ultrasound-mediated direct functionalization of exosomes achieved a high loading efficiency and sustained drug release, surpassing the quantitative effects of electroporation and co-incubation [80]. After sonication treatment of exosomes and berberine, the drug loading reached 17.13%±1.64%, and the cumulative release amount of loaded sample reached 71.44±2.86% within 48 h, thus alleviating inflammation and cell apoptosis caused by SCI [81]. (6) Freeze and thaw cycles encompass three subsequent cycles: incubation at room temperature, rapid freezing at -80 ℃ or in liquid nitrogen, and thawing at room temperature. The phospholipid bilayer of exosomes can bind to liposomes enabling the load of molecules with a high capacity through freeze-thawing methods. For verification, conductive polypyrrole nanoparticles were encapsulated in liposomes and fused with MSCs-derived exosomes to facilitate nerve regeneration via electrical stimulation [82]. (7) Liposome-based transfection involves the fusion of liposomes carrying exogenous genes with exosomes [83]. (8) Proteins on exosomes membrane have abundant amino and alkyl groups that allow for their modification with biomacromolecules through covalent interactions. Click chemistry exhibits the attributes of rapidity, high efficiency and enhanced control over the conjugation site. Hosseini Shamili et al. modified exosomes with carboxylic acid-functionalized aptamers using EDC/NHS chemistry for application in neural myelination [84].
Loading exogenous cargos into exosomes via physical and biochemical approaches, including incubation of exosomes with cargos and membrane permeabilizer, extrusion, electroporation, sonication, freeze and thaw cycles, liposome-based transfection, and click chemistry. Exosomes produced by direct functionalization were used for theranostics, such as biological imaging, drug delivery, and targeting cells. Figures created with Biorender.com.
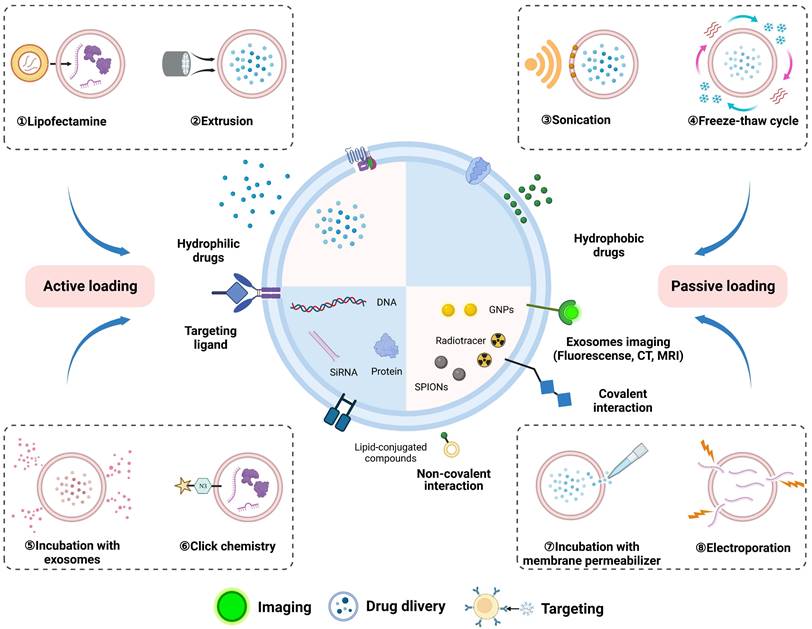
While these engineering techniques help exosomes to load exogenous cargoes for imaging, drug delivery, and targeting, their disadvantages cannot be ignored in the process of application, which include exosomes aggregation, loss of membrane integrity, toxicity, hemolytic activity, unspecific release of drugs [75]. These challenges highlight the need for careful consideration and appropriate strategies to optimize the use of exosomes as theranostic vehicles.
Systematic and local delivery of exosomes in vivo
As non-living nanovesicles, exosomes can be integrated into multiple biomaterials without nutritional support. Considering the structural and functional properties of the PNS and CNS, tailored delivery strategies are necessary to achieve safe, targeted, and sustained therapeutic effects of exosomes. In this context, this section discusses the importance of optimized delivery routes, including intravascular injection, hydrogels, nerve conduits, and 3D printed scaffolds, to facilitate the efficient transportation of exosomes to specific sites within the nervous system (Figure 4).
Approaches for exosomes delivery to the injured sites in peripheral nerve, spinal cord, and brain via local application (direct injection, hydrogel, nerve conduit, and 3D printed scaffolds) and systematic delivery. Figures created with Biorender.com.
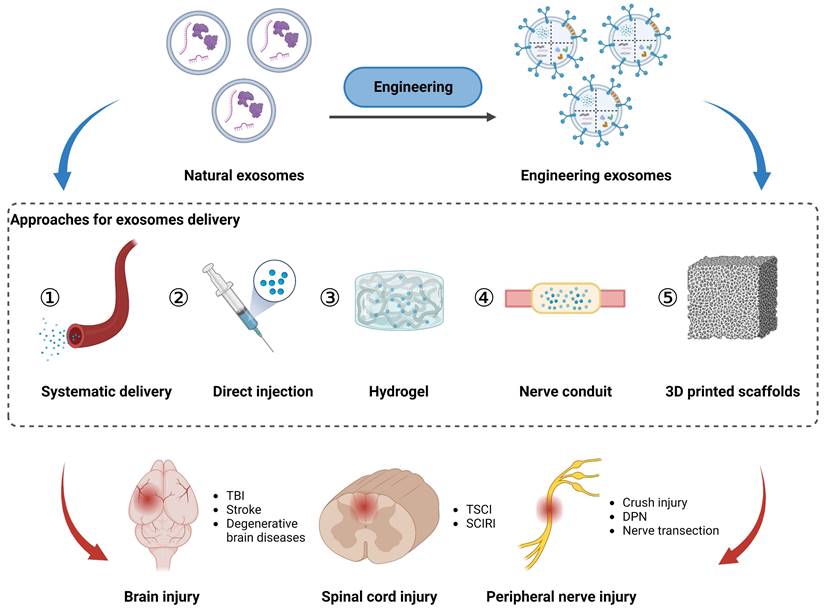
Systematic delivery of exosomes via intravascular injection
Systematic delivery of exosomes using a syringe and needle after mixing them with low- concentration PBS or normal saline is the most therapeutic approach for treating nerve injury via intravascular injection. Several researches have demonstrated the efficacy of injecting exosomes into animal models of spinal cord injury via the tail vein. For instance, Peng et al. injected exosomes derived from the microglial cells into mouse model of traumatic spinal cord injury [85]. They observed that the exosomes began to perform their functions as early as 7 days after surgery, along with significant proliferation of endothelial cells, recovery of neurological function, and a reduction of reactive oxygen species (ROS) levels in the injured area. In vitro, endothelial cells can rapidly internalize microglial-derived exosomes within 12 h, reducing ROS production in H2O2-induced bEnd.3 cells and promoting the proliferation, migration, and tube formation by activating the keap1/Nrf2/HO-1 signaling pathway. Fan et al. reported a similar administration route for delivering exosomes derived from the BMSCs into a rat model of spinal cord injury and observed therapeutic effects after consecutive tail vein injections for 7 days [86]. These experimental results indicate that exosomes can serve as drug nanocarriers, cross biological barriers, and target damaged tissues through systematic applications. However, it is unreasonable to inject exosomes into blood vessels at high concentrations and high frequencies, because the delivery route may cause cytotoxicity of exosomes and local infection at the injection site [87]. Moreover, a large number of exosomes tend to accumulate in the liver, spleen, and lungs through intravascular injection, where they undergo metabolic clearance, thereby limiting the number of exosomes transported to the injured sites [88]. Therefore, seeking the localized maintenance and diffusion of exosomes as a substitute for the injection-based route is a favorable strategy for nerve injury repair.
Hydrogel-mediated sustained release of exosomes
Hydrogels are widely used in regenerative medicine owing to their ability to mimic the extracellular matrix and provide a favorable environment. Encapsulating exosomes within hydrogels offers a way to address the limitations of both exosomes and hydrogels. The incorporation of exosomes into the hydrogel network allows for extended release and enhanced utilization of the exosomes, while the hydrogel acquires the bioactivity from the parent cells. Li et al. combined exosomes from the MSCs with the adhesive peptide PPFLMLLKGSTR-modified hyaluronic acid (HA) hydrogel (Exo-pGel) to repair spinal cord injury lasting more than 4 weeks in rodents, and they observed that more than 90% of the exosomes exhibited sustained release from the 3D pGel for approximately 11 days in vitro [89].
These results may be attributed to the viscosity of HA, which facilitates the slow release of exosomes, and their inherent reparative effects at the nerve injury sites. However, in view of the rapid degradation and slow solidification of HA in vivo, HA hydrogel is unsuitable for engineering applications in severe spinal cord injury. Luo et al. proposed a solution to address the shortcomings of HA [90]. They constructed a photocrosslinkable hydrogel composed of a specific ratio of GelMA, HA-NB, and photo-initiator (LAP) (GelMA/HA-NB), along with exosomes derived from macrophages, for spinal cord injury recovery. After 28 days of soaking in PBS, the hydrogel released 70% of the accumulated exosomes. Moreover, its injection into the spinal cord injured area significantly promoted blood vessel regeneration and functional recovery, which was related to the presence of OTULIN protein in exosomes, activating the Wnt/β-catenin signaling pathway. On the other hand, Han et al. mixed exosomes from 3D-cultured MSCs with GelMA solution and photo-initiator to synthesize a blend solution for exploring the effect of microneedle array patches on spinal cord injury, which was then cast into a nanowell array mold to form GelMA-MN@3D-Exo square patches [91]. The GelMA-MN@3D-Exo patches were evaluated for spinal cord injury that demonstrated great reduction of the inflammatory response, played a neuroprotective role, and reversed the glial/fibrotic scar formation resulting from the excessive aggregation of astrocytes.
Integration of exosomes with other biomaterials
Parallel to hydrogels, various biomaterials have been developed in combination with exosomes for nerve repair. For sciatic nerve dissection injuries, many studies have demonstrated the necessity of bridging between the proximal and distal axons using nerve conduits [92]. Currently, chitosan-based biodegradable nerve conduits are used in clinical treatment, and studies have shown that combining chitosan conduits with exosomes holds great therapeutic potential for peripheral nerve regeneration. Rao et al. successfully repaired sciatic nerve injury by incorporating exosomes derived from gingival MSCs into chitosan conduits. However, exogenous exosomes may leak out of the conduit and rapidly degrade in vivo [93]. To effectively and sustainably harness the therapeutic potential of exosomes, a dopamine-modified chitosan conduit loaded with BMSCs-derived exosomes can stably release exosomes to promote the functional recovery of damaged sciatic nerve [94]. Additionally, engineered exosomes enriched with neurotrophic factor gene have been developed, which can be stably released for at least 2 weeks when mixed with an alginate hydrogel [95]. Rat ADMSCs were successfully loaded with the NT-3 gene sequence through virus-mediated transfection, resulting in exosomes derived from modified MSCs exhibiting high expression of NT-3 mRNA. These NT-3-overexpressed exosomes, combined with alginate hydrogel, were injected into the silicone conduit to assess their therapeutic effects on a 10 mm sciatic nerve defect in vivo. The results showed significant nerve regeneration and functional recovery of the gastrocnemius muscles after 8 weeks. However, the silicone conduit chosen for this study was not degradable and had a high risk of infection and nerve compression in the later stages. With the development of 3D bioprinting in regenerative medicine, 3D printed scaffolds leverage their excellent porosity and interpore channel structure to provide an encapsulation space for exosomes, enabling their controlled release, while also supporting the adhesion of neighboring cells and tissues to the scaffold [96]. We have listed recent reports on exosome-incorporated biomaterials in Table 3 and discuss them extensively in the section on the application of exosomes in peripheral and central nerve injuries. Although the therapeutic potential of exosomes is undeniable, further exploration is required for their clinical application in nerve repair to achieve long-term and localized treatment at the injured sites.
Recent studies using exosomes biomaterials for nerve injury repair in animal Models
| Biomaterials | Exosomes source | Species | Disease | Biological function | Reference |
|---|---|---|---|---|---|
| Chitin conduit | GMSCs | Rat | Sciatic nerve defect | Increased nerve fiber regeneration and myelinization | [93] |
| Polydopamine-modified Chitin conduit | BMSCs | Rat | Sciatic nerve defect | Promoted Schwann cells proliferation and nerve regeneration | [94] |
| Silicone tube with exosomes-alginate hydrogel | ADMSCs | Rat | Sciatic nerve defect | Promoted nerve regeneration and maintained muscle morphology | [95] |
| rGO-GelMA-PCL conduit | BMSCs | Rat | Sciatic nerve defect | Enhanced angiogenesis and axon growth | [97] |
| Core-shell silk-fibroin/PLLA nerve conduit | EnSCs | Rat | Sciatic nerve defect | Enhanced axons regeneration, improved functional recovery | [98] |
| Hyaluronic acid methacrylate hydrogel | UCMSCs | Rat | Sciatic nerve crush injury | Exosome-loaded soft hydrogel promoted nerve repair and anti-inflammation | [99] |
| Alginate scaffold | UCMSCs | Rat | Spinal nerve ligation pain | Antinociceptive, anti-inflammatory, and pro-neurotrophic effects | [100] |
| Peptide-modified adhesive hydrogel | Placenta amniotic membrane MSCs | Rat | Spinal cord injury | Promoted spinal cord regeneration and protected urinary tissue by antioxidation and anti-inflammation | [89] |
| PPFLMLLKGSTR-modified HA hydrogel | UCMSCs | Rat | Spinal cord injury | Replenished spinal cavity, enhanced angiogenesis and locomotor function recovery | [32] |
| Fibrin gel | BMSCs | Mouse | Spinal cord injury | Upregulated neural markers, promoted nerve regeneration and oligodendrogenesis by VGF | [101] |
| Fibrin gel | UCMSCs | Rat | Spinal cord injury | Relieved inflammation and oxidative damage, protected urinary function | [102] |
| Gelatin methacrylate hydrogel | BMSCs | Rat | Spinal cord injury | Promoted neurogenesis, attenuated glial scars | [103] |
| F127-polycitrate-polyethyleneimine hydrogel | ADMSCs | Rat | Spinal cord injury | Suppressed scar formation, reduced inflammatory reaction, promoted remyelination and axonal regeneration. | [104] |
| Gelatin methacrylate/ polypyrrole electroconductive hydrogel | BMSCs | Mouse | Spinal cord injury | Anti-inflammation, enhanced neural stem cells differentiation and axonal growth, inhibited astrocyte differentiation | [105] |
| GelMA hybrid microneedle array patch | MSCs | Rat | Spinal cord injury | Reduced SCI-induced inflammation and glial scarring | [91] |
| Polydopamine-modified hydrogel | Flos sophorae immaturus | Rat | Spinal cord injury | Reduced inflammation and oxidative damage, promoted motor and urinary function | [106] |
| Hyaluronan-collagen hydrogel | BMSCs | Rat | Traumatic brain injury | Induced angiogenesis and neurogenesis, promoted axon growth, remyelination, and synapse formation | [107] |
| Nano-SDF scaffold | NSCs | Rat | Traumatic brain injury | Decreased oxidative stress, reactive gliosis, and neuroinflammation, increased neurogenesis | [108] |
| 3D-printed collagen/silk fibroin scaffold | UCMSCs | Canine | Traumatic brain injury | Inhibited nerve cell apoptosis and proinflammatory factors expression, enhanced motor function recovery | [109] |
| 3D-printed collagen/chitosan scaffold | UCMSCs | Rat | Traumatic brain injury | Recovered neuromotor and cognitive function, improved the regeneration of nerve fibers, synaptic connections, and myelinization | [50] |
| Low-temperature 3D-printed collagen/chitosan scaffold | NSCs | Rat | Traumatic brain injury | Improved motor and cognitive functions, recovered damaged nerve tissue | [110] |
Application of exosomes in peripheral and central nerve injuries
To date, numerous studies have reported the theranostic potential of exosomes in the PNS and CNS. Notably, the exosomes used in these studies were either loaded with different exogenous cargos or exerted their effects on recipient cells via different delivery routes. In this context, the review provides an overview of the applications of exosomes in neuroimaging and emphasizes their comprehensive applications in peripheral nerve, traumatic spinal cord, and traumatic brain injuries.
Neuroimaging
Engineering modifications of exosomes have expanded, changed, and enhanced their therapeutic potential and facilitated neuroimaging. To track exosomes in vivo, various strategies have been developed to effectively label exosomes (Figure 5). Fluorescence imaging, photoacoustic imaging (PAI), CT imaging, MRI imaging, and nuclear imaging (SPECT, PET) are used to non-invasively monitor the absorption, distribution, metabolism and excretion of exosomes. These techniques provide substantial support in elucidating the role of exosomes in the diagnosis and treatment of neurological diseases.
Application of exosomes in imaging and tracking. (A) Labeling strategies of exosomes as natural carriers for diagnosis and treatment. (B) CT imaging of MSCs-derived exosomes using gold nanoparticles as labeling agents. Adapted with permission from [114], copyright 2017, American Chemical Society. (C) Exosomes were loaded with SPIONs and curcumin by electroporation for MRI imaging and therapy of glioma. Adapted with permission from [113], copyright 2018, Elsevier. (D) 99mTc-HMPAO was chosen as a proper radiolabeling of exosome-mimetic nanovesicles (ENVS) to show high brain uptake by SPECT/CT imaging. Adapted with permission from [115], copyright 2015 Springer Nature.
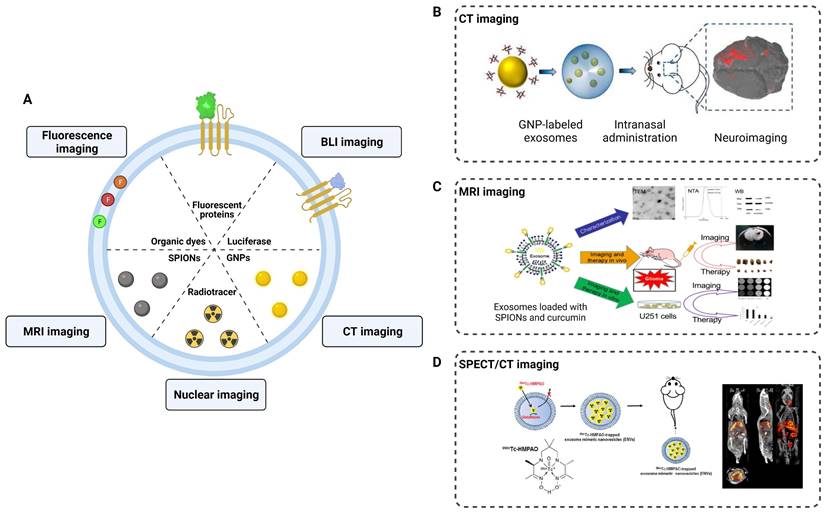
Fluorescence imaging is a commonly employed technique in neuroimaging studies of exosomes. By labeling the surface of exosomes with fluorescent probes, researchers can observe and track the distribution of exosomes in injured nerves. For instance, Bucan et al. used PKH26-labeled exosomes to track the uptake of exosomes by the Schwann cells in vitro and explored their distribution in injured sciatic nerves [111]. However, traditional fluorescence imaging has limitations in terms of tissue penetration depth and fluorescence decay, which hinder the tracking of exosomes in the central nervous system. The blood-brain barrier (BBB) is the primary obstacle for most contrast agents and drugs to enter the brain. Studies have demonstrated that exosomes can traverse the BBB, offering significant prospects for the diagnosis and treatment of CNS diseases. Perets et al. employed GNPs-labeled BMSCs-derived exosomes and performed CT imaging to elucidate their migration and homing patterns in various brain diseases (stroke, autism, Parkinson's and Alzheimer's disease) [112]. The results indicated that the BMSCs-derived exosomes were selectively taken up and accumulated by the neuronal cells in the pathological areas, whereas only diffusive migration with subsequent clearance within 24 h was observed in the control group. Compared to CT imaging, MRI provides higher tissue resolution and avoids radiation exposure in the nervous system. Jia et al. loaded superparamagnetic iron oxide nanoparticles (SPIONs) into exosomes conjugated with myelin basic protein-1 targeting peptide and encapsulated curcumin within the exosomes, achieving targeted MRI imaging and therapy for brain gliomas [113]. In addition, 3D images and the distribution of radionuclide-labeled exosomes in organs can be obtained through SPECT or PET detection. When combined with CT or MRI in hybrid devices, accurate localization of labeled exosomes can be achieved for diagnostic purposes.
Engineering modifications of exosomes provide assurance for the development of neuroimaging techniques. In the future, nuclear imaging, which enables the accurate localization of lesions, along with radiation-free MRI imaging combined with the targeting properties of exosomes, can provide high-resolution structural information for the diagnosis of PNS and CNS diseases.
Peripheral nerve injury
Peripheral nerve injury (PNI) is a complex and prevalent neurological disorder that affects the nerve fibers, supporting cells (Schwann cells), and blood vessels in the PNS. The Schwann cells are involved in the formation of the peripheral nerve myelin sheath, and studies have revealed that exosomes can act directly on the Schwann cells, facilitating peripheral nerve regeneration. In previous study, ADMSCs-derived exosomes promoted the proliferation, migration, myelination, and secretion of SCs, while also inhibiting cell autophagy [63, 116]. Moreover, exosomes derived from the BMSCs combined with PDA-modified chitosan conduit significantly increased the number and diameter of the nerve fibers. This combination promoted myelination by influencing the proliferation, secretion, and reprogramming of the Schwann cells [94]. Interestingly, exosomes secreted by the Schwann cells themselves also contributed to nerve repair. Direct injection of exosomes derived from normoxia-conditioned SCs has been found to promote myelination of the sciatic nerve. However, in vitro experiments have shown that exosomes originating from oxygen-glucose-deprivation-condition SCs (OGD-SCs-Exos) enhanced M1 polarization, thereby limiting axon regeneration and functional recovery (Figure 6A) [117].
Application of exosomes in peripheral nerve injury. (A) OGD-SC-Exos induced the mRNA expression of M1 macrophage markers (IL-6, IL-1β, and TNF-α) in vivo and inhibited the neurites elongation of DRG neurons in vitro. Adapted with permission from [117], copyright 2023, Elsevier. (B) Exosomes derived from LPS-preconditioned MSCs promoted the shift of pro-inflammation macrophage into M2 macrophage phenotype through TSG-6 carried by exosomes inhibiting NF-κB/NLRP3 signaling pathway. Adapted with permission from [120], copyright 2022, Elsevier. (C) Exosomes derived from differentiated hADMSCs with the SCs phenotype attenuated the intracellular oxidative stress of primary SCs and enhanced the tube formation, migration of HUVECs. Adapted with permission from [122], copyright 2021, Elsevier.
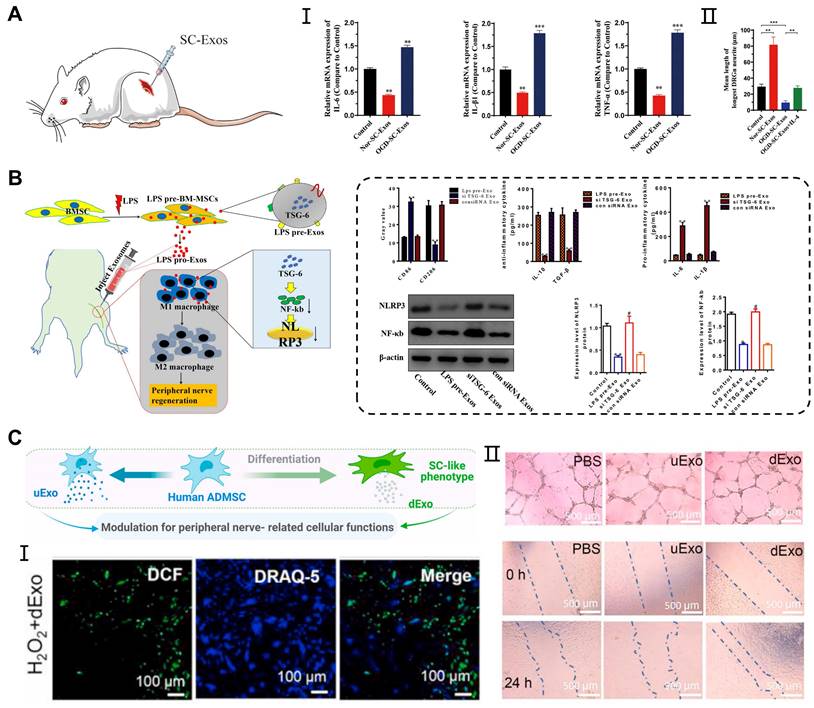
During Wallerian degeneration in peripheral nerve injury, macrophages and other immune cells are recruited to the injured sites to promote the clearance of myelin and axonal debris [118]. Additionally, the reconstruction of the vascular network can provide an appropriate microenvironment for the regeneration and functional recovery of peripheral nerves [119]. Exosomes not only act on the Schwann cells, but also participate in neuroinflammatory responses, oxidative stress, and angiogenesis, contributing to peripheral nerve repair. Experimental studies have shown that local injection of exosomes derived from MSCs pretreated with LPS in a sciatic nerve injury model can induce the transformation of M1 macrophages into M2 polarization, thereby enhancing functional recovery, axon regeneration, and remyelination. RT-qPCR and western blot analysis showed that pretreated exosomes containing TSG-6 contributed to the downregulation of the NF-κB/NLRP3 signaling pathway [120] (Figure 6B). On the other hand, exosomes derived from differentiated hADMSCs with the SCs phenotype (dExo) significantly increased angiogenesis and reduced oxidative stress in peripheral nerve regeneration, which was attributed to several upregulated miRNAs within dExo [121] (Figure 6C). In peripheral nerve injuries, exosome-mediated intercellular communication between neurons and adjacent cells plays an important role in promoting nerve repair. By surface modification and drug loading into exosomes, we can achieve targeted effects of exosomes on peripheral nerve injury through systematic delivery.
Traumatic spinal cord injury
Traumatic spinal cord injury (TSCI) commonly results from in traffic or recreational accidents and often leads to severe neurological dysfunction in the absence of effective treatment [123]. In the past, high-dose methylprednisolone pulse therapy was clinically used to treat acute TSCI; however, its effectiveness was limited by serious side effects [124]. The novel treatment strategy showed that exosomes can be delivered to the spinal cord through intravenous injection, nasal administration, and local application, thereby circumventing the immunogenicity and tumorigenicity of stem cells transplantation [125]. Studies have shown that administration of MSC-derived exosomes via tail vein injection into TSCI mice results in significant injury repair, similar to the restorative effects of MSCs transplantation, but without the accompanying drawbacks associated with cell transplantation [126]. Additionally, Guo et al. found that exosomes carrying siRNA targeting the phosphatase and tensin homolog (PTEN) reduced its expression in the spinal cord injury area following intranasal delivery [127]. The engineered exosomes significantly promoted axon growth and angiogenesis while reducing the proliferation of microglia and astrocytes. However, some exosomes are metabolized by other organs in the bloodstream, thereby limiting their arrival at the injured site and impeding their therapeutic effects (Figure 7A). Studies have shown that early local administration of exosomes can regulate acute-phase inflammation; however, direct and systemic delivery of exosomes has certain limitations.
Application of exosomes in traumatic spinal cord injury. (A) Intranasal delivery of MSCs-derived exosomes crossed the biological barrier to treat spinal cord injury. Quantitative analysis of gold nanoparticle-labeled exosomes from the CNS and organs in healthy and injured rats. Adapted with permission from [127], copyright 2019, American Chemical Society. (B) Reduction in oxidative damage, improvement in M2 macrophages polarization and bladder tissues after fibrin gel-exosomes treatment. Adapted with permission from [102], copyright 2021, Elsevier. (C) Mechanisms of exosomes in GMPE hydrogels modulating inflammatory response and axon growth. Adapted with permission from [105], copyright 2022, Wiley-VCH GmbH.
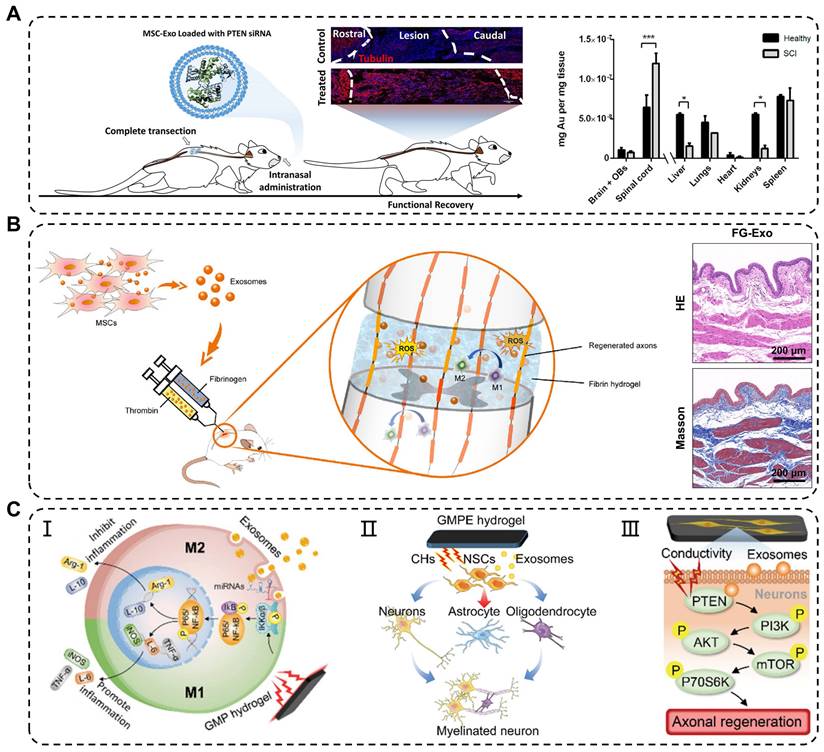
To overcome these limitations, engineering approaches have been developed to combine exosomes with biomaterials, specifically hydrogels, to achieve effective and sustained delivery of exosomes. In one study, Mu et al. reported fibrin gel encapsulation of hUCMSCs-derived exosomes (FG-Exo) for emergency treatment of acute spinal cord injury in rats [102]. The results showed that FG-Exo effectively alleviated oxidative stress and inflammatory response in the spinal cord-injured microenvironment, promoted nerve tissue regeneration, and significantly improved urinary system function (Figure 7B). In another study, Li et al. modified HA hydrogel with laminin-derived peptide (PPFLMLLKGSTR) to create highly adhesive hydrogel (pGel) that retained human placental MSCs-derived exosomes (Exo-pGel) in a severe SCI model [89]. However, natural hydrogels have limited mechanical strength and unstable structures, which restrict their long-term release of exosomes. Synthetic hydrogels, such as GelMA and PLGA hydrogels, are composite biomaterials that offer a balance between biocompatibility and mechanical properties required for spinal cord application. Using 3D GelMA as a delivery system for exosomes (GelMA-Exo) can fill the spinal cord cavity and achieve sustained release. In vitro, GelMA-Exo promoted the survival and neural differentiation of NSCs, reduced glial scar formation, increased axon growth, and effectively promoted the recovery of neurological functions in SCI [103]. FAN et al. developed a dual-network electroconductive hydrogel (GMP) consisting of photocrosslinking GelMA hydrogel and polypyrrole (PPy) hydrogel. MSCs-derived exosomes were incorporated into the GMP network through reversible non-covalent binding to form the GelMA/PPy/Exosomes hydrogel (GMPE) [105]. The application of the GMPE hydrogel to bridge the transected spinal cord resulted in the partial restoration of endogenous electrical signal transmission. Moreover, exosomes released from the GMPE hydrogel can last up to 14 days because the hydrogel has excellent biological adhesion. Implantation of the GMPE hydrogel into the injured spinal cord regulated the polarization of microglia from M1 to M2 through the NF-κB pathway. It also enhanced differentiation of NSCs into neurons and oligodendrocytes and activated the PTEN/PI3K/AKT/mTOR pathway to promote axon growth, synergistically facilitating the recovery of motor function at an early stage of SCI (Figure 7C).
Besides the aforementioned biomaterials, self-assembling peptides (SAPs) have been used to repair spinal cord injury [128]. Therefore, the strategies of biomaterials for exosomes loading provide promising guidance for clinical practice in the treatment of CNS diseases.
Traumatic brain injury
Traumatic brain injury (TBI) is caused by structural damage and/or functional impairment of brain tissue due to external trauma, and is the leading cause of morbidity and mortality among young people worldwide [129]. Severe TBI can be diagnosed on the basis of clinical presentation and imaging observations. However, the existing molecular and imaging examinations for chronic TBI have certain limitations. Hemphill et al. developed a nanofluidic immunomagnetic technique to purify exosomes and analyze their miRNAs [130]. The analysis was performed using chips and machine learning, with a set of biomarkers as indicators for TBI grading and diagnosis (Figure 8A). In the experiments, the technique achieved a correct recognition rate of 99% in both traumatic and sham-operated mice. The role of natural exosomes is limited in the treatment of brain disease. Increasing evidence suggests that appropriate engineering methods can enhance the therapeutic effects of exosomes in traumatic brain injury with neural damage and functional impairments. Wang et al. investigated the effects of genetically modified exosomes on cell apoptosis and neural function following traumatic brain injury [131]. They transfected plasmids carrying Bcl-2 cDNA and Bax shRNA into exosomes derived from astrocytes and then injected functional exosomes into the ventricle after 1 h of surgery. Following 48 h of exosomes treatment, the Mcl-1, XIAP, and Survivin protein levels in the brain were attenuated and the release of cytochrome c from the mitochondria to the cytoplasm was reduced. Moreover, the engineered exosomes mitigated the damage to miniature excitatory postsynaptic currents (mEPSCs) and long-term potentiation (LTP) in the hippocampus of TBI mice, leading to improvement in motor and cognitive behavior.
Application of exosomes in traumatic brain injury. (A) Machine learning-based TBI diagnostics to analyze brain-derived exosomes using immunomagnetic isolation. Adapted with permission from [130], copyright 2018, Royal Society of Chemistry. (B) Exosomes encapsulated in hyaluronan-based hydrogel promoted TBI repair by NSCs differentiation, neurogenesis, and angiogenesis. Adapted with permission from [107], copyright 2023, Elsevier. (C) 3D-CC-BMExo increased the number of positive cells for nerve fibers, synaptic connections, and myelin regeneration at implanted site. Adapted with permission from [50], copyright 2022, Oxford University Press.
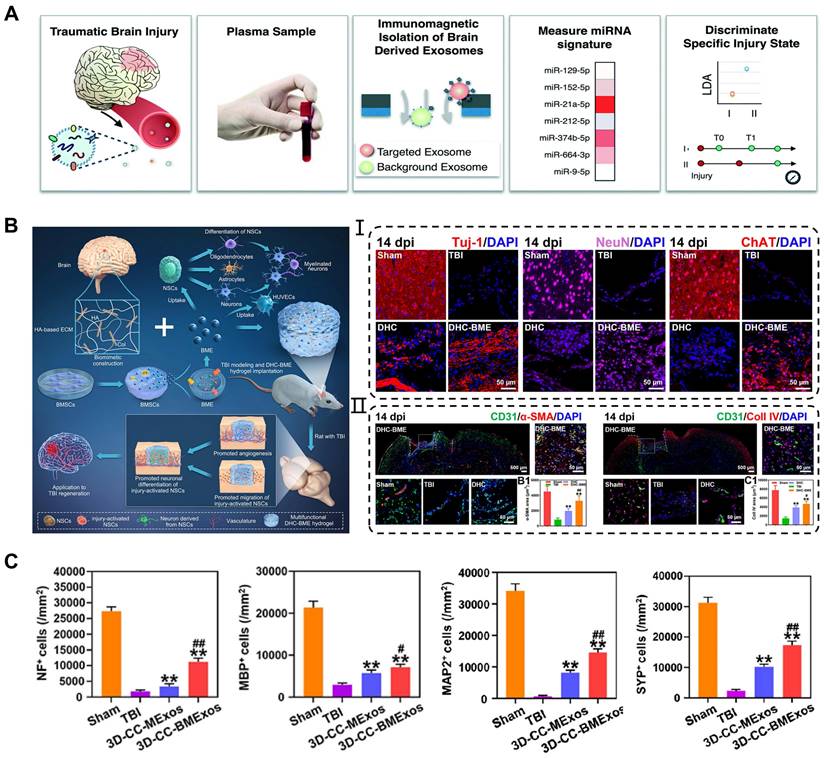
Although exosomes can be administered intravenously or injected directly for TBI, the application of sustained-release and defect-filling biomaterials optimized their delivery route. Liu et al. combined BMSCs-derived exosomes with HA-collagen hydrogel to simulate the brain matrix and achieved a stable release of exosomes [107]. These results revealed that the hydrogel encapsulated exosomes induced angiogenesis and neurogenesis at the injured site, consequently promoting neural function recovery after TBI (Figure 8B). Recently, many researchers have successfully integrated bioscaffolds and exosomes using 3D printing technology to attain the desired mechanical properties and biocompatibility of biomaterials for TBI treatment. Liu et al. reported that combining 3D printed collagen/chitosan scaffolds with BDNF-pretreated hUCMSCs-derived exosomes (3D-CC-BMExos) enhanced the reparative effects of exosomes on TBI [50]. The bioscaffold exhibited promising biocompatibility and degradation properties, and the incorporation of 3D-CC-BMExo further promoted neural network remodeling by improving nerve fibers, synaptic connections, and myelin regeneration at the implanted site (Figure 8C). Similarly, the research team treated TBI in a beagle model by combining 3D printed collagen/silk fibroin scaffolds with exosomes obtained from hypoxia-pretreated hUCMSCs (3D-CSHMExos) and found that 3D-CSHMExos significantly promoted neural regeneration and angiogenesis. Furthermore, 3D-CSHMExos further inhibited neuronal apoptosis and pro-inflammatory cytokines (TNF-α and IL-6) while increasing the expression of anti-inflammatory cytokine (IL-10), ultimately facilitating motor function recovery after TBI [109]. In this review, we provided a concise overview of the optimized application of exosomes in TBI. However, current research is still confined to the animal models, necessitating extensive investigation for clinical applications.
Conclusion and Perspectives
Neurological disorders can arise from various causes, such as tumors, autoimmunity, and genetic factors, aside from direct damage to the axons and cell bodies of the peripheral and central nerves. Traditional therapeutic strategies only provide symptomatic relief on a macroscopic level, without fully restoring neural structure or removing risk factors. Exosomes, with nanomembrane structures, have the ability to effortlessly cross the BBB, blood-spinal cord barrier, and blood-nerve barrier while maintaining their biological activity. These features present a promising therapeutic and diagnostic strategy for neurological diseases, for which extensive research has been conducted on the mechanisms, delivery pathways, and production improvement. Exosomes carry a variety of molecules on their surface and interior, including integrins, ceramides, MHC, proteins, RNA, and DNA; however, not all properties can be fully utilized in the application process. Thus, exosomes need to be functionalized for specific purposes, such as targeting, bioimaging, and drug loading. Advancement in cell engineering, genetic engineering, and biomaterials technology have provided technical support for these specific applications of exosomes. Enhancing our understanding of the biogenesis of exosomes by regulating parental cell culture parameters and utilizing various bioreactors have improved exosomes production for basic research; however, current isolation techniques greatly restricted their clinical potential.
The focus of exosomes research has progressively shifted towards clinical applications for the treatment of various diseases. Chu et al. utilized exosomes derived from MSCs for nebulized therapy in seven COVID-19 patients, contributing to improving the absorption of lung lesions and reducing hospitalization time for mild cases of COVID-19 [132]. However, there are still no reports of clinical trials investigating the use of exosomes for nerve injury repair (source: ClinicalTrials.gov). In order to provide researchers with guidance on producing exosomes in accordance with good manufacturing practices, the Minimal Information for Studies of Extracellular Vesicles (MISEV) from the International Society for Extracellular Vesicles (ISEV) has provided extensive information and instructions [133]. Nevertheless, there is no detailed explanation of the safety issues related to the surface modification and gene loading of exosomes, and the potential side effects in clinical applications remain unclear. Moreover, the clinical application of exosomes raises ethical and safety concerns owing to the challenges associated with the cell source for exosomes secretion, particularly in the context of allogeneic sources, and the potential contamination of extracted exosomes with other proteins, which has not been fully demonstrated in clinical trials. Even when using MSCs from autologous sources, meeting the high demand for exosomes remains challenging, especially considering the criticality of early treatment for nerve injury. Hence, mechanical stimulation and dynamic culture system are necessary to promote exosomes production for the preparation of therapeutic reagents. However, these parameters must be strictly adjusted to avoid the overexpression of inflammatory factors and non-exosomes vesicle shedding. Various biological repair materials have been widely applied in clinical treatment, ensuring their safety and stability, but further research is needed to explore their combined application with exosomes for the effective treatment of neurological diseases. Despite being undeniably crucial for nerve injury repair, exosomes are relatively novel drug used in the field of tissue engineering. Therefore, further research on the clinical efficacy and production of exosomes is necessary before their safe application in humans.
Abbreviations
ADMSCs: adipose-derived mesenchymal stem cells; BMSCs: bone mesenchymal stem cells; BNDF: brain-derived neurotrophic factor; CNS: central nervous system; CTGF: connective tissue growth factor; COVID-19: corona virus disease 2019; DRG: dorsal root ganglion; EnSCs: endometrial stem cells; ESCRT: endosomal sorting complex required for transport; EVs: extracellular vesicles; GelMA: gelatin methacryloyl; GMSCs: gingival mesenchymal stem cells; GNPs: gold nanoparticles; HA: hyaluronic acid; HUVECs: human umbilical vein endothelial cells; HIF-1α: hypoxia-inducible 1-alpha; IGF-1: insulin-like growth factor 1; ISEV: international society for extracellular vesicles; ILVs: intraluminal vesicles; KDR: kinase insert domain receptor; LTP: long-term potentiation; LIPUS: low-intensity pulsed ultrasound; MRI: magnetic resonance imaging; MSCs: mesenchymal stem cells; MCAO: middle cerebral artery occlusion; mEPSCs: miniature excitatory postsynaptic currents; MISEV: minimal information for studies of extracellular vesicles; MVBs: multivesicular bodies; MDSCs: muscle-derived stem cells; NSCs: neural stem cells; OGD/R: oxygen-glucose deprivation and reperfusion; PNS: peripheral nervous system; PNI: peripheral nerve injury; PBS: phosphate buffered solution; PLGA: poly(lactic-co-glycolic acid); PLLA: poly-L-lactic acid; PET: positron emission tomography; ROS: reactive oxygen species; SCs: Schwann cells; SPECT: single-photon emission computed tomography; SPIONs: superparamagnetic iron oxide nanoparticles; TFF: tangential flow filtration; TBI: traumatic brain injury; TSCI: traumatic spinal cord injury; UCMSCs: umbilical cord mesenchymal stem cells; VEGFA: vascular endothelial growth factor A.
Acknowledgements
This work was supported by the Natural Foundation of Beijing Municipality [Grant No.7222198], Peking University People's Hospital Research and Development Funds [NO. RDX2021-01].
Competing Interests
The authors have declared that no competing interest exists.
References
1. Wüthrich T, Lese I, Haberthür D, Zubler C, Hlushchuk R, Hewer E. et al. Development of vascularized nerve scaffold using perfusion-decellularization and recellularization. Mater Sci Eng C Mater Biol Appl. 2020;117:111311
2. Mahar M, Cavalli V. Intrinsic mechanisms of neuronal axon regeneration. Nat Rev Neurosci. 2018;19:323-37
3. Kaminska A, Radoszkiewicz K, Rybkowska P, Wedzinska A, Sarnowska A. Interaction of Neural Stem Cells (NSCs) and Mesenchymal Stem Cells (MSCs) as a Promising Approach in Brain Study and Nerve Regeneration. Cells. 2022 11
4. Bell JH, Haycock JW. Next generation nerve guides: materials, fabrication, growth factors, and cell delivery. Tissue Eng Part B Rev. 2012;18:116-28
5. Harilal S, Jose J, Parambi DGT, Kumar R, Unnikrishnan MK, Uddin MS. et al. Revisiting the blood-brain barrier: A hard nut to crack in the transportation of drug molecules. Brain Res Bull. 2020;160:121-40
6. Todorova D, Simoncini S, Lacroix R, Sabatier F, Dignat-George F. Extracellular Vesicles in Angiogenesis. Circ Res. 2017;120:1658-73
7. Qing L, Chen H, Tang J, Jia X. Exosomes and Their MicroRNA Cargo: New Players in Peripheral Nerve Regeneration. Neurorehabil Neural Repair. 2018;32:765-76
8. Huang J, Zhang G, Li S, Li J, Wang W, Xue J. et al. Endothelial cell-derived exosomes boost and maintain repair-related phenotypes of Schwann cells via miR199-5p to promote nerve regeneration. J Nanobiotechnology. 2023;21:10
9. Chen J, Zhang C, Li S, Li Z, Lai X, Xia Q. Exosomes Derived from Nerve Stem Cells Loaded with FTY720 Promote the Recovery after Spinal Cord Injury in Rats by PTEN/AKT Signal Pathway. J Immunol Res. 2021;2021:8100298
10. Hade MD, Suire CN, Suo Z. Mesenchymal Stem Cell-Derived Exosomes: Applications in Regenerative Medicine. Cells. 2021 10
11. Gebeyehu A, Kommineni N, Meckes DG Jr, Sachdeva MS. Role of Exosomes for Delivery of Chemotherapeutic Drugs. Crit Rev Ther Drug Carrier Syst. 2021;38:53-97
12. Yuan J, Botchway BOA, Zhang Y, Wang X, Liu X. Combined bioscaffold with stem cells and exosomes can improve traumatic brain injury. Stem Cell Rev Rep. 2020;16:323-34
13. Johnstone RM, Adam M, Hammond JR, Orr L, Turbide C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262:9412-20
14. Lu Y, Mai Z, Cui L, Zhao X. Engineering exosomes and biomaterial-assisted exosomes as therapeutic carriers for bone regeneration. Stem Cell Res Ther. 2023;14:55
15. Liang Y, Duan L, Lu J, Xia J. Engineering exosomes for targeted drug delivery. Theranostics. 2021;11:3183-95
16. Gurung S, Perocheau D, Touramanidou L, Baruteau J. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal. 2021;19:47
17. Harrison EB, Hochfelder CG, Lamberty BG, Meays BM, Morsey BM, Kelso ML. et al. Traumatic brain injury increases levels of miR-21 in extracellular vesicles: implications for neuroinflammation. FEBS Open Bio. 2016;6:835-46
18. Long X, Yao X, Jiang Q, Yang Y, He X, Tian W. et al. Astrocyte-derived exosomes enriched with miR-873a-5p inhibit neuroinflammation via microglia phenotype modulation after traumatic brain injury. J Neuroinflammation. 2020;17:89
19. Huang S, Ge X, Yu J, Han Z, Yin Z, Li Y. et al. Increased miR-124-3p in microglial exosomes following traumatic brain injury inhibits neuronal inflammation and contributes to neurite outgrowth via their transfer into neurons. FASEB J. 2018;32:512-28
20. Frühbeis C, Kuo-Elsner WP, Müller C, Barth K, Peris L, Tenzer S. et al. Oligodendrocytes support axonal transport and maintenance via exosome secretion. PLoS Biol. 2020;18:e3000621
21. Lopez-Leal R, Court FA. Schwann Cell Exosomes Mediate Neuron-Glia Communication and Enhance Axonal Regeneration. Cell Mol Neurobiol. 2016;36:429-36
22. Zhan C, Ma CB, Yuan HM, Cao BY, Zhu JJ. Macrophage-derived microvesicles promote proliferation and migration of Schwann cell on peripheral nerve repair. Biochem Biophys Res Commun. 2015;468:343-8
23. Yang Y, Ye Y, Su X, He J, Bai W, He X. MSCs-Derived Exosomes and Neuroinflammation, Neurogenesis and Therapy of Traumatic Brain Injury. Front Cell Neurosci. 2017;11:55
24. Zeng X, Bian W, Liu Z, Li J, Ren S, Zhang J. et al. Muscle-derived stem cell exosomes with overexpressed miR-214 promote the regeneration and repair of rat sciatic nerve after crush injury to activate the JAK2/STAT3 pathway by targeting PTEN. Front Mol Neurosci. 2023;16:1146329
25. Yu T, Xu Y, Ahmad MA, Javed R, Hagiwara H, Tian X. Exosomes as a Promising Therapeutic Strategy for Peripheral Nerve Injury. Curr Neuropharmacol. 2021;19:2141-51
26. Liang Y, Wu JH, Zhu JH, Yang H. Exosomes Secreted by Hypoxia-Pre-conditioned Adipose-Derived Mesenchymal Stem Cells Reduce Neuronal Apoptosis in Rats with Spinal Cord Injury. J Neurotrauma. 2022;39:701-14
27. Liu X, Zhang M, Liu H, Zhu R, He H, Zhou Y. et al. Bone marrow mesenchymal stem cell-derived exosomes attenuate cerebral ischemia-reperfusion injury-induced neuroinflammation and pyroptosis by modulating microglia M1/M2 phenotypes. Exp Neurol. 2021;341:113700
28. Wang ZG, He ZY, Liang S, Yang Q, Cheng P, Chen AM. Comprehensive proteomic analysis of exosomes derived from human bone marrow, adipose tissue, and umbilical cord mesenchymal stem cells. Stem Cell Res Ther. 2020;11:511
29. Schnyder SK, Molina JJ, Yamamoto R. Control of cell colony growth by contact inhibition. Sci Rep. 2020;10:6713
30. Patel DB, Gray KM, Santharam Y, Lamichhane TN, Stroka KM, Jay SM. Impact of cell culture parameters on production and vascularization bioactivity of mesenchymal stem cell-derived extracellular vesicles. Bioeng Transl Med. 2017;2:170-9
31. Dou L, Yan Q, Liang P, Zhou P, Zhang Y, Ji P. iTRAQ-Based Proteomic Analysis Exploring the Influence of Hypoxia on the Proteome of Dental Pulp Stem Cells under 3D Culture. Proteomics. 2018 18
32. Mu J, Li L, Wu J, Huang T, Zhang Y, Cao J. et al. Hypoxia-stimulated mesenchymal stem cell-derived exosomes loaded by adhesive hydrogel for effective angiogenic treatment of spinal cord injury. Biomater Sci. 2022;10:1803-11
33. Abdollahi S. Extracellular vesicles from organoids and 3D culture systems. Biotechnol Bioeng. 2021;118:1029-49
34. Haraszti RA, Miller R, Stoppato M, Sere YY, Coles A, Didiot MC. et al. Exosomes Produced from 3D Cultures of MSCs by Tangential Flow Filtration Show Higher Yield and Improved Activity. Mol Ther. 2018;26:2838-47
35. Xu R, Zhang F, Chai R, Zhou W, Hu M, Liu B. et al. Exosomes derived from pro-inflammatory bone marrow-derived mesenchymal stem cells reduce inflammation and myocardial injury via mediating macrophage polarization. J Cell Mol Med. 2019;23:7617-31
36. Harting MT, Srivastava AK, Zhaorigetu S, Bair H, Prabhakara KS, Toledano Furman NE. et al. Inflammation-Stimulated Mesenchymal Stromal Cell-Derived Extracellular Vesicles Attenuate Inflammation. Stem Cells. 2018;36:79-90
37. Wu D, Kang L, Tian J, Wu Y, Liu J, Li Z. et al. Exosomes Derived from Bone Mesenchymal Stem Cells with the Stimulation of Fe(3)O(4) Nanoparticles and Static Magnetic Field Enhance Wound Healing Through Upregulated miR-21-5p. Int J Nanomedicine. 2020;15:7979-93
38. Kim HY, Kim TJ, Kang L, Kim YJ, Kang MK, Kim J. et al. Mesenchymal stem cell-derived magnetic extracellular nanovesicles for targeting and treatment of ischemic stroke. Biomaterials. 2020;243:119942
39. Jia L, Chopp M, Wang L, Lu X, Szalad A, Zhang ZG. Exosomes derived from high-glucose-stimulated Schwann cells promote development of diabetic peripheral neuropathy. FASEB J. 2018;32:fj201800597R
40. Lv H, Li L, Sun M, Zhang Y, Chen L, Rong Y. et al. Mechanism of regulation of stem cell differentiation by matrix stiffness. Stem Cell Res Ther. 2015;6:103
41. Shah N, Morsi Y, Manasseh R. From mechanical stimulation to biological pathways in the regulation of stem cell fate. Cell Biochem Funct. 2014;32:309-25
42. Zhong W, Tian K, Zheng X, Li L, Zhang W, Wang S. et al. Mesenchymal stem cell and chondrocyte fates in a multishear microdevice are regulated by Yes-associated protein. Stem Cells Dev. 2013;22:2083-93
43. Kimiz-Gebologlu I, Oncel SS. Exosomes: Large-scale production, isolation, drug loading efficiency, and biodistribution and uptake. J Control Release. 2022;347:533-43
44. Yan L, Wu X. Exosomes produced from 3D cultures of umbilical cord mesenchymal stem cells in a hollow-fiber bioreactor show improved osteochondral regeneration activity. Cell Biol Toxicol. 2020;36:165-78
45. de Almeida Fuzeta M, Bernardes N, Oliveira FD, Costa AC, Fernandes-Platzgummer A, Farinha JP. et al. Scalable Production of Human Mesenchymal Stromal Cell-Derived Extracellular Vesicles Under Serum-/Xeno-Free Conditions in a Microcarrier-Based Bioreactor Culture System. Front Cell Dev Biol. 2020;8:553444
46. Ye K, Li Z, Yin Y, Zhou J, Li D, Gan Y. et al. LIPUS-SCs-Exo promotes peripheral nerve regeneration in cavernous nerve crush injury-induced ED rats via PI3K/Akt/FoxO signaling pathway. CNS Neurosci Ther. 2023
47. Wang C, Ji Y, Zhang H, Ye Y, Zhang G, Zhang S. et al. Increased level of exosomal miR-20b-5p derived from hypothermia-treated microglia promotes neurite outgrowth and synapse recovery after traumatic brain injury. Neurobiol Dis. 2023: 106042.
48. Wang Y, Yu T, Hu F. Hypocapnia Stimuli-Responsive Engineered Exosomes Delivering miR-218 Facilitate Sciatic Nerve Regeneration. Front Bioeng Biotechnol. 2022;10:825146
49. Ma K, Xu H, Zhang J, Zhao F, Liang H, Sun H. et al. Insulin-like growth factor-1 enhances neuroprotective effects of neural stem cell exosomes after spinal cord injury via an miR-219a-2-3p/YY1 mechanism. Aging (Albany NY). 2019;11:12278-94
50. Liu X, Zhang J, Cheng X, Liu P, Feng Q, Wang S. et al. Integrated printed BDNF-stimulated HUCMSCs-derived exosomes/collagen/chitosan biological scaffolds with 3D printing technology promoted the remodelling of neural networks after traumatic brain injury. Regen Biomater. 2023;10:rbac085
51. Xu H, Jia Z, Ma K, Zhang J, Dai C, Yao Z. et al. Protective effect of BMSCs-derived exosomes mediated by BDNF on TBI via miR-216a-5p. Med Sci Monit. 2020;26:e920855
52. Rau CS, Kuo PJ, Wu SC, Huang LH, Lu TH, Wu YC. et al. Enhanced Nerve Regeneration by Exosomes Secreted by Adipose-Derived Stem Cells with or without FK506 Stimulation. Int J Mol Sci. 2021 22
53. Kuo PJ, Rau CS, Wu SC, Lin CW, Huang LH, Lu TH. et al. Exosomes Secreted by Adipose-Derived Stem Cells Following FK506 Stimulation Reduce Autophagy of Macrophages in Spine after Nerve Crush Injury. Int J Mol Sci. 2021 22
54. Pusic AD, Kraig RP. Youth and environmental enrichment generate serum exosomes containing miR-219 that promote CNS myelination. Glia. 2014;62:284-99
55. Pusic KM, Pusic AD, Kraig RP. Environmental Enrichment Stimulates Immune Cell Secretion of Exosomes that Promote CNS Myelination and May Regulate Inflammation. Cell Mol Neurobiol. 2016;36:313-25
56. Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9:654-9
57. Zhang J, Li S, Li L, Li M, Guo C, Yao J. et al. Exosome and exosomal microRNA: trafficking, sorting, and function. Genomics Proteomics Bioinformatics. 2015;13:17-24
58. Liang ZY, Xu XJ, Rao J, Yang ZL, Wang CH, Chen CM. Mesenchymal Stem Cell-Derived Exosomal MiRNAs Promote M2 Macrophages Polarization: Therapeutic Opportunities for Spinal Cord Injury. Front Mol Neurosci. 2022;15:926928
59. Wang X, Zhou Y, Gao Q, Ping D, Wang Y, Wu W. et al. The Role of Exosomal microRNAs and Oxidative Stress in Neurodegenerative Diseases. Oxid Med Cell Longev. 2020;2020:3232869
60. Li D, Zhang P, Yao X, Li H, Shen H, Li X. et al. Exosomes Derived From miR-133b-Modified Mesenchymal Stem Cells Promote Recovery After Spinal Cord Injury. Front Neurosci. 2018;12:845
61. Chang Q, Hao Y, Wang Y, Zhou Y, Zhuo H, Zhao G. Bone marrow mesenchymal stem cell-derived exosomal microRNA-125a promotes M2 macrophage polarization in spinal cord injury by downregulating IRF5. Brain Res Bull. 2021;170:199-210
62. Yang J, Wang B, Wang Y, Feng C, Chen L, Liu Y. et al. Exosomes Derived from Adipose Mesenchymal Stem Cells Carrying miRNA-22-3p Promote Schwann Cells Proliferation and Migration through Downregulation of PTEN. Dis Markers. 2022;2022:7071877
63. Yin G, Yu B, Liu C, Lin Y, Xie Z, Hu Y. et al. Exosomes produced by adipose-derived stem cells inhibit schwann cells autophagy and promote the regeneration of the myelin sheath. Int J Biochem Cell Biol. 2021;132:105921
64. Wang Y, Lai X, Wu D, Liu B, Wang N, Rong L. Umbilical mesenchymal stem cell-derived exosomes facilitate spinal cord functional recovery through the miR-199a-3p/145-5p-mediated NGF/TrkA signaling pathway in rats. Stem Cell Res Ther. 2021;12:117
65. Zhou Z, Li C, Bao T, Zhao X, Xiong W, Luo C. et al. Exosome-Shuttled miR-672-5p from Anti-Inflammatory Microglia Repair Traumatic Spinal Cord Injury by Inhibiting AIM2/ASC/Caspase-1 Signaling Pathway Mediated Neuronal Pyroptosis. J Neurotrauma. 2022;39:1057-74
66. Ren ZW, Zhou JG, Xiong ZK, Zhu FZ, Guo XD. Effect of exosomes derived from MiR-133b-modified ADSCs on the recovery of neurological function after SCI. Eur Rev Med Pharmacol Sci. 2019;23:52-60
67. Li R, Zhao K, Ruan Q, Meng C, Yin F. Bone marrow mesenchymal stem cell-derived exosomal microRNA-124-3p attenuates neurological damage in spinal cord ischemia-reperfusion injury by downregulating Ern1 and promoting M2 macrophage polarization. Arthritis Res Ther. 2020;22:75
68. Li D, Huang S, Yin Z, Zhu J, Ge X, Han Z. et al. Increases in miR-124-3p in Microglial Exosomes Confer Neuroprotective Effects by Targeting FIP200-Mediated Neuronal Autophagy Following Traumatic Brain Injury. Neurochem Res. 2019;44:1903-23
69. Li D, Huang S, Zhu J, Hu T, Han Z, Zhang S. et al. Exosomes from MiR-21-5p-Increased Neurons Play a Role in Neuroprotection by Suppressing Rab11a-Mediated Neuronal Autophagy In vitro After Traumatic Brain Injury. Med Sci Monit. 2019;25:1871-85
70. Wen L, Wang YD, Shen DF, Zheng PD, Tu MD, You WD. et al. Exosomes derived from bone marrow mesenchymal stem cells inhibit neuroinflammation after traumatic brain injury. Neural Regen Res. 2022;17:2717-24
71. Ding W, Gu Q, Liu M, Zou J, Sun J, Zhu J. Astrocytes-derived exosomes pre-treated by berberine inhibit neuroinflammation after stroke via miR-182-5p/Rac1 pathway. Int Immunopharmacol. 2023;118:110047
72. Zhang Y, Chopp M, Liu XS, Katakowski M, Wang X, Tian X. et al. Exosomes Derived from Mesenchymal Stromal Cells Promote Axonal Growth of Cortical Neurons. Mol Neurobiol. 2017;54:2659-73
73. Xin H, Katakowski M, Wang F, Qian JY, Liu XS, Ali MM. et al. MicroRNA cluster miR-17-92 Cluster in Exosomes Enhance Neuroplasticity and Functional Recovery After Stroke in Rats. Stroke. 2017;48:747-53
74. Luan X, Sansanaphongpricha K, Myers I, Chen H, Yuan H, Sun D. Engineering exosomes as refined biological nanoplatforms for drug delivery. Acta Pharmacol Sin. 2017;38:754-63
75. Peng H, Ji W, Zhao R, Yang J, Lu Z, Li Y. et al. Exosome: a significant nano-scale drug delivery carrier. J Mater Chem B. 2020;8:7591-608
76. He R, Jiang Y, Shi Y, Liang J, Zhao L. Curcumin-laden exosomes target ischemic brain tissue and alleviate cerebral ischemia-reperfusion injury by inhibiting ROS-mediated mitochondrial apoptosis. Mater Sci Eng C Mater Biol Appl. 2020;117:111314
77. Haney MJ, Klyachko NL, Zhao Y, Gupta R, Plotnikova EG, He Z. et al. Exosomes as drug delivery vehicles for Parkinson's disease therapy. J Control Release. 2015;207:18-30
78. Khongkow M, Yata T, Boonrungsiman S, Ruktanonchai UR, Graham D, Namdee K. Surface modification of gold nanoparticles with neuron-targeted exosome for enhanced blood-brain barrier penetration. Sci Rep. 2019;9:8278
79. Huang W, Qu M, Li L, Liu T, Lin M, Yu X. SiRNA in MSC-derived exosomes silences CTGF gene for locomotor recovery in spinal cord injury rats. Stem Cell Res Ther. 2021;12:334
80. Kim MS, Haney MJ, Zhao Y, Mahajan V, Deygen I, Klyachko NL. et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine. 2016;12:655-64
81. Gao ZS, Zhang CJ, Xia N, Tian H, Li DY, Lin JQ. et al. Berberine-loaded M2 macrophage-derived exosomes for spinal cord injury therapy. Acta Biomater. 2021;126:211-23
82. Singh A, Raghav A, Shiekh PA, Kumar A. Transplantation of engineered exosomes derived from bone marrow mesenchymal stromal cells ameliorate diabetic peripheral neuropathy under electrical stimulation. Bioact Mater. 2021;6:2231-49
83. Cheng X, Zhou H, Zhou Y, Song C. M2 Macrophage-Derived Exosomes Inhibit Apoptosis of HUVEC Cell through Regulating miR-221-3p Expression. Biomed Res Int. 2022;2022:1609244
84. Hosseini Shamili F, Alibolandi M, Rafatpanah H, Abnous K, Mahmoudi M, Kalantari M. et al. Immunomodulatory properties of MSC-derived exosomes armed with high affinity aptamer toward mylein as a platform for reducing multiple sclerosis clinical score. J Control Release. 2019;299:149-64
85. Peng W, Wan L, Luo Z, Xie Y, Liu Y, Huang T. et al. Microglia-Derived Exosomes Improve Spinal Cord Functional Recovery after Injury via Inhibiting Oxidative Stress and Promoting the Survival and Function of Endothelia Cells. Oxid Med Cell Longev. 2021;2021:1695087
86. Fan L, Dong J, He X, Zhang C, Zhang T. Bone marrow mesenchymal stem cells-derived exosomes reduce apoptosis and inflammatory response during spinal cord injury by inhibiting the TLR4/MyD88/NF-κB signaling pathway. Hum Exp Toxicol. 2021;40:1612-23
87. Bei HP, Hung PM, Yeung HL, Wang S, Zhao X. Bone-a-Petite: Engineering Exosomes towards Bone, Osteochondral, and Cartilage Repair. Small. 2021;17:e2101741
88. Wiklander OP, Nordin JZ, O'Loughlin A, Gustafsson Y, Corso G, Mäger I. et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;4:26316
89. Li L, Zhang Y, Mu J, Chen J, Zhang C, Cao H. et al. Transplantation of Human Mesenchymal Stem-Cell-Derived Exosomes Immobilized in an Adhesive Hydrogel for Effective Treatment of Spinal Cord Injury. Nano Lett. 2020;20:4298-305
90. Luo Z, Peng W, Xu Y, Xie Y, Liu Y, Lu H. et al. Exosomal OTULIN from M2 macrophages promotes the recovery of spinal cord injuries via stimulating Wnt/β-catenin pathway-mediated vascular regeneration. Acta Biomater. 2021;136:519-32
91. Han M, Yang H, Lu X, Li Y, Liu Z, Li F. et al. Three-Dimensional-Cultured MSC-Derived Exosome-Hydrogel Hybrid Microneedle Array Patch for Spinal Cord Repair. Nano Lett. 2022;22:6391-401
92. Fang X, Deng J, Zhang W, Guo H, Yu F, Rao F. et al. Conductive conduit small gap tubulization for peripheral nerve repair. RSC Adv. 2020;10:16769-75
93. Rao F, Zhang D, Fang T, Lu C, Wang B, Ding X. et al. Exosomes from Human Gingiva-Derived Mesenchymal Stem Cells Combined with Biodegradable Chitin Conduits Promote Rat Sciatic Nerve Regeneration. Stem Cells Int. 2019;2019:2546367
94. Li C, Liu SY, Zhang M, Pi W, Wang B, Li QC. et al. Sustained release of exosomes loaded into polydopamine-modified chitin conduits promotes peripheral nerve regeneration in rats. Neural Regen Res. 2022;17:2050-7
95. Yang Z, Yang Y, Xu Y, Jiang W, Shao Y, Xing J. et al. Biomimetic nerve guidance conduit containing engineered exosomes of adipose-derived stem cells promotes peripheral nerve regeneration. Stem Cell Res Ther. 2021;12:442
96. Poongodi R, Chen YL, Yang TH, Huang YH, Yang KD, Lin HC. et al. Bio-Scaffolds as Cell or Exosome Carriers for Nerve Injury Repair. Int J Mol Sci. 2021 22
97. Zhang W, Fang XX, Li QC, Pi W, Han N. Reduced graphene oxide-embedded nerve conduits loaded with bone marrow mesenchymal stem cell-derived extracellular vesicles promote peripheral nerve regeneration. Neural Regen Res. 2023;18:200-6
98. Namini MS, Ebrahimi-Barough S, Ai J, Jahromi HK, Mikaeiliagah E, Azami M. et al. Tissue-Engineered Core-Shell Silk-Fibroin/Poly-l-Lactic Acid Nerve Guidance Conduit Containing Encapsulated Exosomes of Human Endometrial Stem Cells Promotes Peripheral Nerve Regeneration. ACS Biomater Sci Eng. 2023;9:3496-511
99. Liu Z, Tong H, Li J, Wang L, Fan X, Song H. et al. Low-Stiffness Hydrogels Promote Peripheral Nerve Regeneration Through the Rapid Release of Exosomes. Front Bioeng Biotechnol. 2022;10:922570
100. Hsu JM, Shiue SJ, Yang KD, Shiue HS, Hung YW, Pannuru P. et al. Locally Applied Stem Cell Exosome-Scaffold Attenuates Nerve Injury-Induced Pain in Rats. J Pain Res. 2020;13:3257-68
101. He X, Yang L, Dong K, Zhang F, Liu Y, Ma B. et al. Biocompatible exosome-modified fibrin gel accelerates the recovery of spinal cord injury by VGF-mediated oligodendrogenesis. J Nanobiotechnology. 2022;20:360
102. Mu J, Wu J, Cao J, Ma T, Li L, Feng S. et al. Rapid and effective treatment of traumatic spinal cord injury using stem cell derived exosomes. Asian J Pharm Sci. 2021;16:806-15
103. Cheng J, Chen Z, Liu C, Zhong M, Wang S, Sun Y. et al. Bone mesenchymal stem cell-derived exosome-loaded injectable hydrogel for minimally invasive treatment of spinal cord injury. Nanomedicine (Lond). 2021;16:1567-79
104. Wang C, Wang M, Xia K, Wang J, Cheng F, Shi K. et al. A bioactive injectable self-healing anti-inflammatory hydrogel with ultralong extracellular vesicles release synergistically enhances motor functional recovery of spinal cord injury. Bioact Mater. 2021;6:2523-34
105. Fan L, Liu C, Chen X, Zheng L, Zou Y, Wen H. et al. Exosomes-Loaded Electroconductive Hydrogel Synergistically Promotes Tissue Repair after Spinal Cord Injury via Immunoregulation and Enhancement of Myelinated Axon Growth. Adv Sci (Weinh). 2022;9:e2105586
106. Chen J, Wu J, Mu J, Li L, Hu J, Lin H. et al. An antioxidative sophora exosome-encapsulated hydrogel promotes spinal cord repair by regulating oxidative stress microenvironment. Nanomedicine. 2023;47:102625
107. Liu X, Wu C, Zhang Y, Chen S, Ding J, Chen Z. et al. Hyaluronan-based hydrogel integrating exosomes for traumatic brain injury repair by promoting angiogenesis and neurogenesis. Carbohydr Polym. 2023;306:120578
108. Hajinejad M, Ebrahimzadeh MH, Ebrahimzadeh-Bideskan A, Rajabian A, Gorji A, Sahab Negah S. Exosomes and Nano-SDF Scaffold as a Cell-Free-Based Treatment Strategy Improve Traumatic Brain Injury Mechanisms by Decreasing Oxidative Stress, Neuroinflammation, and Increasing Neurogenesis. Stem Cell Rev Rep. 2023
109. Liu X, Wang J, Wang P, Zhong L, Wang S, Feng Q. et al. Hypoxia-pretreated mesenchymal stem cell-derived exosomes-loaded low-temperature extrusion 3D-printed implants for neural regeneration after traumatic brain injury in canines. Front Bioeng Biotechnol. 2022;10:1025138
110. Liu XY, Feng YH, Feng QB, Zhang JY, Zhong L, Liu P. et al. Low-temperature 3D-printed collagen/chitosan scaffolds loaded with exosomes derived from neural stem cells pretreated with insulin growth factor-1 enhance neural regeneration after traumatic brain injury. Neural Regen Res. 2023;18:1990-8
111. Bucan V, Vaslaitis D, Peck CT, Strauß S, Vogt PM, Radtke C. Effect of Exosomes from Rat Adipose-Derived Mesenchymal Stem Cells on Neurite Outgrowth and Sciatic Nerve Regeneration After Crush Injury. Mol Neurobiol. 2019;56:1812-24
112. Perets N, Betzer O, Shapira R, Brenstein S, Angel A, Sadan T. et al. Golden Exosomes Selectively Target Brain Pathologies in Neurodegenerative and Neurodevelopmental Disorders. Nano Lett. 2019;19:3422-31
113. Jia G, Han Y, An Y, Ding Y, He C, Wang X. et al. NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials. 2018;178:302-16
114. Betzer O, Perets N, Angel A, Motiei M, Sadan T, Yadid G. et al. In vivo Neuroimaging of Exosomes Using Gold Nanoparticles. ACS Nano. 2017;11:10883-93
115. Hwang DW, Choi H, Jang SC, Yoo MY, Park JY, Choi NE. et al. Noninvasive imaging of radiolabeled exosome-mimetic nanovesicle using (99m)Tc-HMPAO. Sci Rep. 2015;5:15636
116. Chen J, Ren S, Duscher D, Kang Y, Liu Y, Wang C. et al. Exosomes from human adipose-derived stem cells promote sciatic nerve regeneration via optimizing Schwann cell function. J Cell Physiol. 2019;234:23097-110
117. Sun J, Liao Z, Li Z, Li H, Wu Z, Chen C. et al. Down-regulation miR-146a-5p in Schwann cell-derived exosomes induced macrophage M1 polarization by impairing the inhibition on TRAF6/NF-κB pathway after peripheral nerve injury. Exp Neurol. 2023;362:114295
118. Zigmond RE, Echevarria FD. Macrophage biology in the peripheral nervous system after injury. Prog Neurobiol. 2019;173:102-21
119. Wang G, Lu P, Qiao P, Zhang P, Cai X, Tang L. et al. Blood vessel remodeling in late stage of vascular network reconstruction is essential for peripheral nerve regeneration. Bioeng Transl Med. 2022;7:e10361
120. Li C, Li X, Shi Z, Wu P, Fu J, Tang J. et al. Exosomes from LPS-preconditioned bone marrow MSCs accelerated peripheral nerve regeneration via M2 macrophage polarization: Involvement of TSG-6/NF-κB/NLRP3 signaling pathway. Exp Neurol. 2022;356:114139
121. Shi Q, Qian Z, Liu D, Sun J, Wang X, Liu H. et al. GMSC-Derived Exosomes Combined with a Chitosan/Silk Hydrogel Sponge Accelerates Wound Healing in a Diabetic Rat Skin Defect Model. Front Physiol. 2017;8:904
122. Liu B, Kong Y, Shi W, Kuss M, Liao K, Hu G. et al. Exosomes derived from differentiated human ADMSC with the Schwann cell phenotype modulate peripheral nerve-related cellular functions. Bioact Mater. 2022;14:61-75
123. Zipser CM, Cragg JJ, Guest JD, Fehlings MG, Jutzeler CR, Anderson AJ. et al. Cell-based and stem-cell-based treatments for spinal cord injury: evidence from clinical trials. Lancet Neurol. 2022;21:659-70
124. Liu Z, Yang Y, He L, Pang M, Luo C, Liu B. et al. High-dose methylprednisolone for acute traumatic spinal cord injury: A meta-analysis. Neurology. 2019;93:e841-e50
125. Abbaszadeh H, Ghorbani F, Derakhshani M, Movassaghpour A, Yousefi M. Human umbilical cord mesenchymal stem cell-derived extracellular vesicles: A novel therapeutic paradigm. J Cell Physiol. 2020;235:706-17
126. Lu Y, Zhou Y, Zhang R, Wen L, Wu K, Li Y. et al. Bone Mesenchymal Stem Cell-Derived Extracellular Vesicles Promote Recovery Following Spinal Cord Injury via Improvement of the Integrity of the Blood-Spinal Cord Barrier. Front Neurosci. 2019;13:209
127. Guo S, Perets N, Betzer O, Ben-Shaul S, Sheinin A, Michaelevski I. et al. Intranasal Delivery of Mesenchymal Stem Cell Derived Exosomes Loaded with Phosphatase and Tensin Homolog siRNA Repairs Complete Spinal Cord Injury. ACS Nano. 2019;13:10015-28
128. Wang Z, Jia S, Xu H, Wang X, Lu B, Wu W. et al. A functionalized self-assembling peptide containing E7 and YIGSR sequences enhances neuronal differentiation of spermatogonial stem cells on aligned PCL fibers for spinal cord injury repair. Theranostics. 2022;12:7567-85
129. Chen Y, Li J, Ma B, Li N, Wang S, Sun Z. et al. MSC-derived exosomes promote recovery from traumatic brain injury via microglia/macrophages in rat. Aging (Albany NY). 2020;12:18274-96
130. Ko J, Hemphill M, Yang Z, Sewell E, Na YJ, Sandsmark DK. et al. Diagnosis of traumatic brain injury using miRNA signatures in nanomagnetically isolated brain-derived extracellular vesicles. Lab Chip. 2018;18:3617-30
131. Wang B, Han S. Modified Exosomes Reduce Apoptosis and Ameliorate Neural Deficits Induced by Traumatic Brain Injury. ASAIO J. 2019;65:285-92
132. Chu M, Wang H, Bian L, Huang J, Wu D, Zhang R. et al. Nebulization Therapy with Umbilical Cord Mesenchymal Stem Cell-Derived Exosomes for COVID-19 Pneumonia. Stem Cell Rev Rep. 2022;18:2152-63
133. Théry C, Witwer KW, Aikawa E, Alcaraz MJ, Anderson JD, Andriantsitohaina R. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J Extracell Vesicles. 2018;7:1535750
Author contact
Corresponding authors: Na Han, Department of Trauma and Orthopedics, Peking University People's Hospital, Beijing 100000, China. E-mail: hannaqacom, hannaqacom; Yuhui Kou, Department of Trauma and Orthopedics, Peking University People's Hospital, Beijing 100000, China. E-mail: yuhuikouedu.cn.
 Global reach, higher impact
Global reach, higher impact