13.3
Impact Factor
Theranostics 2015; 5(9):931-945. doi:10.7150/thno.11802 This issue Cite
Research Paper
Smart MoS2/Fe3O4 Nanotheranostic for Magnetically Targeted Photothermal Therapy Guided by Magnetic Resonance/Photoacoustic Imaging
1. Key Laboratory of Polymer Science and Technology, School of Science, Northwestern Polytechnical University, Xi'an, Shaanxi, China
2. CAS Key Laboratory for Biomedical Effects of Nanomaterials and Nanosafety, Institute of High Energy Physics, Beijing, China
3. School of Life Science and Technology, Xidian University, Xi'an, Shaanxi, China
#Jie Yu and Wenyan Yin contributed equally to this study.
Abstract
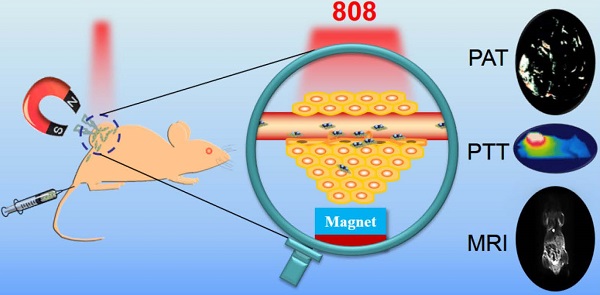
The ability to selectively destroy cancer cells while sparing normal tissue is highly desirable during the cancer therapy. Here, magnetic targeted photothermal therapy was demonstrated by the integration of MoS2 (MS) flakes and Fe3O4 (IO) nanoparticles (NPs), where MoS2 converted near-infrared (NIR) light into heat and Fe3O4 NPs served as target moiety directed by external magnetic field to tumor site. The MoS2/Fe3O4 composite (MSIOs) functionalized by biocompatible polyethylene glycol (PEG) were prepared by a simple two-step hydrothermal method. And the as-obtained MSIOs exhibit high stability in bio-fluids and low toxicity in vitro and in vivo. Specifically, the MSIOs can be applied as a dual-modal probe for T2-weighted magnetic resonance (MR) and photoacoustic tomography (PAT) imaging due to their superparamagnetic property and strong NIR absorption. Furthermore, we demonstrate an effective result for magnetically targeted photothermal ablation of cancer. All these results show a great potential for localized photothermal ablation of cancer spatially/timely guided by the magnetic field and indicated the promise of the multifunctional MSIOs for applications in cancer theranostics.
Keywords: Magnetic target, Photothermal therapy, Magnetic resonance imaging, Photoacoustic tomography imaging, Theranostic.
 Global reach, higher impact
Global reach, higher impact