Impact Factor
Theranostics 2015; 5(11):1264-1274. doi:10.7150/thno.11848 This issue Cite
Research Paper
Biomimetic Acoustically-Responsive Vesicles for Theranostic Applications
Department of Biomedical Engineering and Environmental Sciences, National Tsing Hua University, Hsinchu, 30013, Taiwan
Abstract
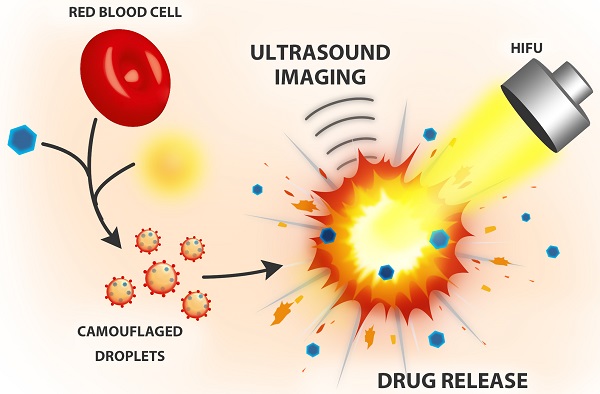
In recent years, biomimetic cell membrane-derived particles have emerged as a new class of drug delivery system with advantages of biocompatibility, ease of isolation and long circulation profile. Here we report the development and potential theranostic applications of a new biomimetic acoustically-responsive droplet system derived from mammalian red blood cell membrane (RBCM). We hypothesized that drug-loaded RBCM droplets (RBCMDs) would undergo a transition from liquid (droplets) to gas (bubbles) upon high intensity focused ultrasound (HIFU) insonation, resulting in on-demand drug release. The generated microbubbles could also serve as a contrast agent to enhance ultrasound imaging. As-synthesized RBCMDs exhibited uniform size, good dispersity and preservation of RBCM-associated proteins that prevented uptake by macrophages. Camptothecin (CPT), an anti-cancer drug, was successfully loaded in the RBCMDs with a loading efficiency of 2-3% and an encapsulation efficiency of 62-97%. A short (3 min) exposure to HIFU irradiation triggered release of CPT from the RBCMDs and the physical explosion of droplets damaged nearby cancer cells resulting in significant cell death. In addition, the acoustically vaporized RBCMDs significantly increased the ultrasound echo signal to 30 dB. Lastly, we demonstrated that RBCMDs could be acoustically vaporized in vivo in target tissues, and enhancing ultrasound imaging. Taken together, we have developed a new class of naturally derived RBCMDs which show great potential for future application in remotely triggered drug delivery and ultrasound imaging enhancement.
Keywords: red blood cell membrane, ultrasound theranostics, remotely triggerable drug delivery, cancer therapy, biomimetic vesicles.
 Global reach, higher impact
Global reach, higher impact