Impact Factor
Theranostics 2017; 7(1):51-66. doi:10.7150/thno.16074 This issue Cite
Research Paper
Localized Delivery of shRNA against PHD2 Protects the Heart from Acute Myocardial Infarction through Ultrasound-Targeted Cationic Microbubble Destruction
1. Department of Ultrasound, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Hubei Province Key Laboratory of Molecular Imaging, Wuhan, China;
2. Paul C. Lauterbur Research Center for Biomedical Imaging, Institute of biomedical and Health Engineering, Shenzhen Institutes of Advanced Technology, Chinese Academy of Sciences, Shenzhen, China.
*Both authors contributed equally to this manuscript.
Abstract
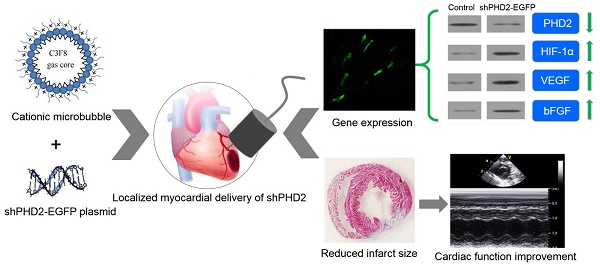
Hypoxia-inducible factor 1α (HIF-1α) plays a critical protective role in ischemic heart disease. Under normoxic conditions, HIF-1α was degraded by oxygen-dependent prolyl hydroxylase-2 (PHD2). Gene therapy has become a promising strategy to inhibit the degradation of HIF-1α and to improve cardiac function after ischemic injury. However, conventional gene delivery systems are difficult to achieve a targeted and localized gene delivery into the ischemic myocardia. Here, we report the localized myocardial delivery of shRNA against PHD2 through ultrasound-targeted microbubble destruction (UTMD) for protection the heart from acute myocardial infarction. In this study, a novel cationic microbubble was fabricated by using of the thin-film hydration and sonication method. The resulting microbubbles had a 28.2 ± 2.21 mV surface zeta potential and could greatly improve DNA binding performance, achieving 17.81 ± 1.46 μg of DNA loading capacity per 5 × 108 microbubbles. Combined with these cationic microbubbles, UTMD-mediated gene delivery was evaluated and the gene transfection efficiency was optimized in the H9C2 cardiac cells. Knockdown of PHD2 gene was successfully realized by UTMD-mediated shPHD2 transfection, resulting in HIF-1α-dependent protective effects on H9C2 cells through increasing the expression of HIF-1α, VEGF and bFGF. We further employed UTMD-mediated shPHD2 transfection into the localized ischemic myocardia in a rat ischemia model, demonstrating significantly reduced infarct size and greatly improved the heart function. The silencing of PHD2 and the up-regulation of its downstream genes in the treated myocardia were confirmed. Histological analysis further revealed numbers of HIF-1α- and VEGF-, and CD31-positive cells/mm2 in the shPHD2-treated group were significantly greater than those in the sham or control vector groups (P < 0.05). In conclusion, our study provides a promising strategy to realize ultrasound-mediated localized myocardial shRNA delivery to protect the heart from acute myocardial infarction via cationic microbubbles.
Keywords: Prolyl hydroxylase-2, Ischemic myocardial disease, Gene delivery, Ultrasound, Cationic microbubbles.
 Global reach, higher impact
Global reach, higher impact