Impact Factor
Theranostics 2017; 7(7):1875-1889. doi:10.7150/thno.18985 This issue Cite
Research Paper
Self-Assembly of Gold Nanoparticles Shows Microenvironment-Mediated Dynamic Switching and Enhanced Brain Tumor Targeting
1. Department of Neurology, Shanghai East Hospital, Tongji University School of Medicine, Shanghai, 200120, China;
2. The Institute for Translational Nanomedicine, Shanghai East Hospital
The Institute for Biomedical Engineering & Nano Science, Tongji University School of Medicine, Shanghai, 200120, China
3. College of Chemistry and Molecular Science, The Institute for Advanced Studies, Wuhan University, Wuhan 430072, P.R. China;
4. Northwestern University Feinberg School of Medicine, 676 North Saint Clair Street, Suite 2210, Chicago, Illinois 60611, United States.
Abstract
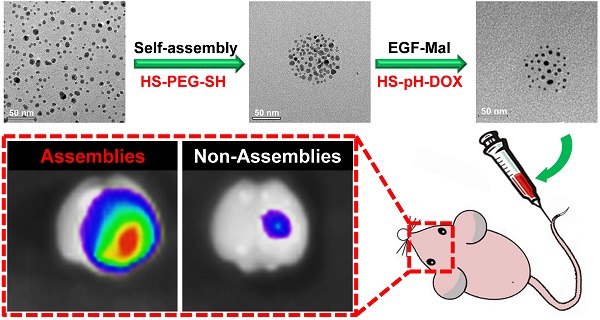
Inorganic nanoparticles with unique physical properties have been explored as nanomedicines for brain tumor treatment. However, the clinical applications of the inorganic formulations are often hindered by the biological barriers and failure to be bioeliminated. The size of the nanoparticle is an essential design parameter which plays a significant role to affect the tumor targeting and biodistribution. Here, we report a feasible approach for the assembly of gold nanoparticles into ~80 nm nanospheres as a drug delivery platform for enhanced retention in brain tumors with the ability to be dynamically switched into the single formulation for excretion. These nanoassemblies can target epidermal growth factor receptors on cancer cells and are responsive to tumor microenvironmental characteristics, including high vascular permeability and acidic and redox conditions. Anticancer drug release was controlled by a pH-responsive mechanism. Intracellular L-glutathione (GSH) triggered the complete breakdown of nanoassemblies to single gold nanoparticles. Furthermore, in vivo studies have shown that nanospheres display enhanced tumor-targeting efficiency and therapeutic effects relative to single-nanoparticle formulations. Hence, gold nanoassemblies present an effective targeting strategy for brain tumor treatment.
Keywords: Self-Assembly, Gold nanoparticles, Tumor microenvironment, L-glutathione (GSH), Blood-brain barrier (BBB).
 Global reach, higher impact
Global reach, higher impact