Impact Factor
Theranostics 2017; 7(9):2363-2376. doi:10.7150/thno.19898 This issue Cite
Research Paper
Quantification of Tumor Vascular Permeability and Blood Volume by Positron Emission Tomography
1. Department of Nuclear Medicine and Minnan PET Center, Xiamen Cancer Hospital, the First Affiliated Hospital of Xiamen University, Xiamen, China;
2. Laboratory of Molecular Imaging and Nanomedicine, National Institute of Biomedical Imaging and Bioengineering, National Institutes of Health, Bethesda, Maryland;
3. Section on Quantitative Imaging & Tissue Science, National Institute of Child Health and Human Development, National Institutes of Health, Bethesda, Maryland.
* These authors contributed equally.
Abstract
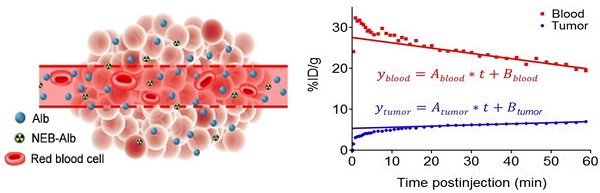
Purpose: Evans Blue (EB) is an azo dye that binds quantitatively with serum albumin. With an albumin binding, NOTA conjugated truncated Evan's blue (NEB) dye derived PET tracer, we aimed to establish a strategy for evaluating vascular permeability in malignant tumors via non-invasive PET. Experimental design: Sixty-minute dynamic PET using [18F]FAl-NEB was performed in three xenograft tumor models including INS-1 rat insulinoma, UM-SCC-22B human head and neck carcinoma and U-87 MG human glioblastoma. Tumor vascular permeability was quantified by the difference of the slopes between tumor and blood time-activity curve (TACs, expressed as Ps). The method was further substantiated by EB extraction and colorimetric assay and correlates with that calculated from dynamic contrast enhanced magnetic resonance imaging (DCE-MRI). The changes in tumor vasculature at different time points were assessed with NEB PET in U-87 MG and UM-SCC-22B tumor models after treatment with bevacizumab or doxorubicin. Result: The Ps values calculated from tumor and blood TACs from multiple time-point static images are consistent with those from dynamic images. Moreover, the Ps showed a positive and significant correlation with extracted EB concentration and KPS-MRI generated from DCE-MRI, which further confirmed the soundness of this methodology. The antiangiogenic effect of bevacizumab could be revealed by NEB PET in U-87 MG tumors as early as 8 hrs after therapy, demonstrated by a substantial decrease of Ps. On the contrary, there was no significant change of Ps in bevacizumab treated UM-SCC-22B tumors, compared with control group. However, the significant changes of Ps were overestimated in doxorubicin treated UM-SCC-22B tumors. Conclusions: We successfully developed a relatively convenient and novel strategy to evaluate vascular permeability and blood volume using NEB PET. This method will be advantageous in evaluating vascular permeability, promoting drug delivery, and monitoring tumor response to therapeutics that affect tumor angiogenesis.
Keywords: Evans blue, positron emission tomography, tumor vasculature, therapy response, vascular permeability.
 Global reach, higher impact
Global reach, higher impact