Impact Factor
Theranostics 2017; 7(16):4001-4012. doi:10.7150/thno.18788 This issue Cite
Research Paper
Ginsenoside Rg1 Inhibits Glucagon-Induced Hepatic Gluconeogenesis through Akt-FoxO1 Interaction
1. State Key Laboratory of Natural Medicines, China Pharmaceutical University, Nanjing 210009, China;
2. Clinical Metabolomics Center, China Pharmaceutical University, Nanjing 211198, China.
* The authors contributed equally to the work.
Abstract
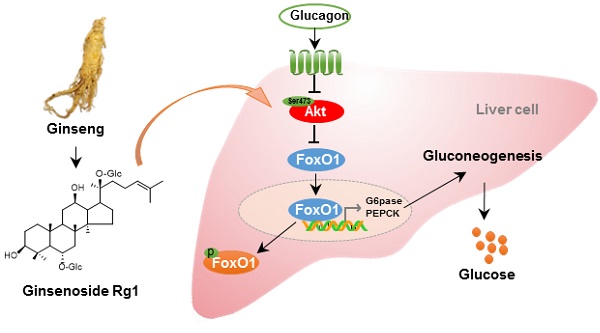
Rationale: Glucagon is involved in hepatic gluconeogenesis, playing a key role in type 2 diabetes. Ginsenosides are reported to have antidiabetic activities. Ginsenoside Rg1 is a major propanaxatriol-type saponin in ginseng. This study aims to investigate the regulatory effects of Rg1 on glucagon-induced hepatic glucose production.
Methods: The effects of Rg1 were investigated in high-fat-diet (HFD)-fed mice and glucagon-challenged C57BL/6J mice. Glucose metabolism was evaluated by oral glucose tolerance test and pyruvate tolerance test. The role of Rg1 on the regulation of Akt-FoxO1 interaction was performed using immunofluorescence, immunoprecipitation, siRNA silencing, pharmacological inhibitor and active-site mutant in primary hepatocytes or HepG2 cells.
Results: Abnormally elevated fasting glucagon levels were observed in HFD-fed mice, contributing significantly to increased fasting plasma glucose levels. Inappropriate fasting glucagon secretion inactivated Akt and promoted hepatic glucose production via upregulation of FoxO1 activity. Rg1 preserved glucagon-impaired Akt activation partly by binding to Akt at Ser473 site. Rg1 also promoted Akt binding to FoxO1 and inactivated FoxO1 by phosphorylation. Consequently, Rg1 decreased the hepatic glucose production through a decrease in transcription of phosphoenolpyruvate carboxykinase (PEPCK) and glucose 6-phosphatase (G6Pase). Both siRNA silencing of Akt and Akt inhibitor triciribine attenuated the effects of Rg1 in response to fasting hormone glucagon.
Conclusion: Akt phosphorylation at Ser473 by ginsenoside Rg1 is critical for its gluconeogenesis-lowering effect, suggesting a potential for pharmaceutical intervention in response to fasting hormone glucagon.
Keywords: ginsenoside Rg1, glucagon, gluconeogenesis, Akt, FoxO1.
 Global reach, higher impact
Global reach, higher impact