Impact Factor
Theranostics 2018; 8(4):1027-1041. doi:10.7150/thno.22414 This issue Cite
Research Paper
Targeting ALDH2 for Therapeutic Interventions in Chronic Pain-Related Myocardial Ischemic Susceptibility
1. Department of Pathophysiology, Fourth Military Medical University, Xi'an, 710032, China;
2. Department of Physiology, Fourth Military Medical University, Xi'an, 710032, China;
3. Fuwai Hospital, National Center of Cardiovascular Diseases, Beijing 100037, China;
4. Department of Cardiovascular Surgery, Xijing Hospital, Fourth Military Medical University, Xi'an, 710032, China;
5. Cardiovascular center, First Affiliated Hospital of Jilin University, Changchun, 130000, China;
6. Department of Pathology, Xijing Hospital, Fourth Military Medical University, Xi'an, 710032, China.
* These authors contributed equally to this work
Abstract
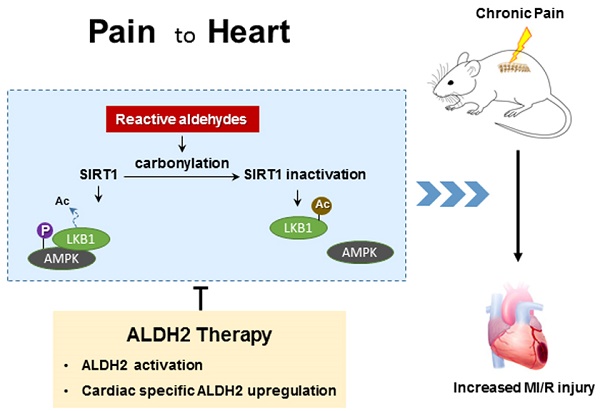
Clinical observations have demonstrated a link between chronic pain and increased ischemic heart disease mortality, but the mechanisms remain elusive. Reactive aldehydes have recently been confirmed as a new player in pain pathologies, while our previous study demonstrated that reactive aldehydes (4-HNE) induced carbonyl stress contributing to myocardial ischemic intolerance. The aim of this study was to explore whether chronic pain increases susceptibility to myocardial ischemia/reperfusion (MI/R) injury and to investigate the underlying mechanisms focusing on toxic aldehyde and carbonyl stress.
Methods: Chronic pain was induced by chronic compression of the dorsal root ganglion (CCD). After 2 weeks CCD, aldehyde dehydrogenase (ALDH2) KO or wild-type (WT) littermate mice were then subjected to in vivo MI/R.
Results: In CCD-WT mice, heightened nociception paralleled circulating aldehyde (4-HNE) accumulation and cardiac protein carbonylation. Mechanistically, CCD-induced 4-HNE overload provoked cardiac Sirtuin 1 (SIRT1) carbonylative inactivation and inhibited Liver kinase B1 (LKB1) - AMP-activated protein kinase (LKB1-AMPK) interaction, which resulted in exacerbated MI/R injury and higher mortality compared with non-CCD WT mice. ALDH2 deficiency further aggravated CCD-induced susceptibility to MI/R injury. Exogenous 4-HNE exposure in peripheral tissue mimicked chronic pain-induced aldehyde overload, elicited sustained allodynia and increased MI/R injury. However, cardiac-specific ALDH2 upregulation by AAV9-cTNT-mediated gene delivery significantly ameliorated chronic pain-induced SIRT1 carbonylative inactivation and decreased MI/R injury (minor infarct size, less apoptosis, and improved cardiac function).
Conclusion: Collectively, chronic pain-enhanced carbonyl stress promotes myocardial ischemic intolerance by SIRT1 carbonylative inactivation and impairment of LKB1-AMPK interaction. ALDH2 activation and prevention of protein carbonylation may be a potential therapeutic target for myocardial ischemic vulnerability in chronic pain patients. Our results newly provided overlapping cellular mechanisms of chronic pain and myocardial dysfunction interplay.
Keywords: chronic pain, myocardial ischemic intolerance, ALDH2, 4-HNE, carbonylation.
 Global reach, higher impact
Global reach, higher impact