Impact Factor
Theranostics 2018; 8(9):2300-2312. doi:10.7150/thno.21670 This issue Cite
Research Paper
Companion Diagnostic 64Cu-Liposome Positron Emission Tomography Enables Characterization of Drug Delivery to Tumors and Predicts Response to Cancer Nanomedicines
1. Merrimack Pharmaceuticals, Inc.
2. TECHNA Institute for the Advancement of Technology for Health, University Health Network, Toronto, Ontario, Canada.
3. Institute of Biomaterials and Biomedical Engineering, University of Toronto, Ontario, Canada.
*Affiliation at the time of study.
Abstract
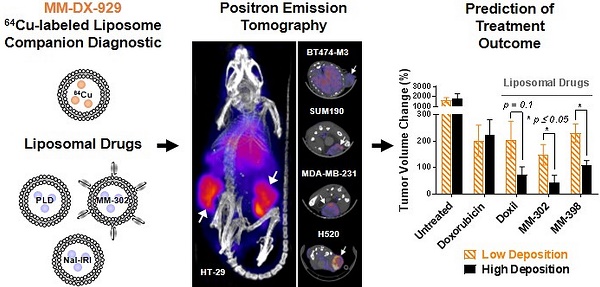
Deposition of liposomal drugs into solid tumors is a potentially rate-limiting step for drug delivery and has substantial variability that may influence probability of response. Tumor deposition is a shared mechanism for liposomal therapeutics such that a single companion diagnostic agent may have utility in predicting response to multiple nanomedicines.
Methods: We describe the development, characterization and preclinical proof-of-concept of the positron emission tomography (PET) agent, MM-DX-929, a drug-free untargeted 100 nm PEGylated liposome stably entrapping a chelated complex of 4-DEAP-ATSC and 64Cu (copper-64). MM-DX-929 is designed to mimic the biodistribution of similarly sized therapeutic agents and enable quantification of deposition in solid tumors.
Results: MM-DX-929 demonstrated sufficient in vitro and in vivo stability with PET images accurately reflecting the disposition of liposome nanoparticles over the time scale of imaging. MM-DX-929 is also representative of the tumor deposition and intratumoral distribution of three different liposomal drugs, including targeted liposomes and those with different degrees of PEGylation. Furthermore, stratification using a single pre-treatment MM-DX-929 PET assessment of tumor deposition demonstrated that tumors with high MM-DX-929 deposition predicted significantly greater anti-tumor activity after multi-cycle treatments with different liposomal drugs. In contrast, MM-DX-929 tumor deposition was not prognostic in untreated tumor-bearing xenografts, nor predictive in animals treated with small molecule chemotherapeutics.
Conclusions: These data illustrate the potential of MM-DX-929 PET as a companion diagnostic strategy to prospectively select patients likely to respond to liposomal drugs or nanomedicines of similar molecular size.
Keywords: 64Cu-liposome PET, nanomedicine, tumor deposition
 Global reach, higher impact
Global reach, higher impact