13.3
Impact Factor
Theranostics 2018; 8(11):3022-3037. doi:10.7150/thno.23046 This issue Cite
Research Paper
PIAS3-mediated feedback loops promote chronic colitis-associated malignant transformation
1. State Key Laboratory of Pharmaceutical Biotechnology, School of Life Sciences, Nanjing University, Nanjing, Jiangsu 210046, China
2. Department of General Surgery, Jinling Hospital, School of Medicine, Nanjing University, Nanjing, Jiangsu 210002, China
3. State Key Laboratory of Analytical Chemistry for Life Sciences and Collaborative Innovation Center of Chemistry for Life Sciences, Nanjing University, Nanjing, Jiangsu 210046, China
Abstract
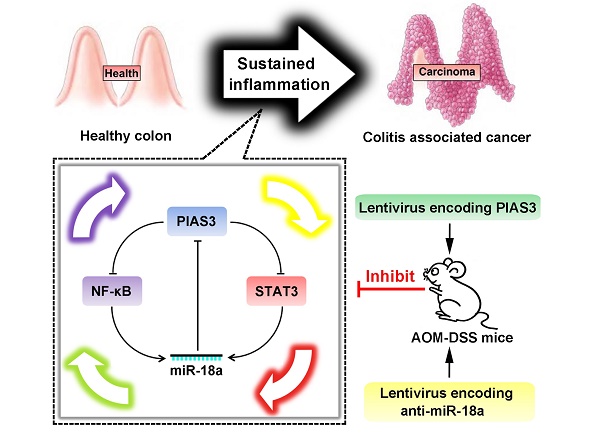
Rationale: Colitis-associated colorectal cancer (CAC) usually exhibits an accelerated disease progression, an increased resistance to therapeutic drugs and a higher mortality rate than sporadic colorectal cancer (CRC). PIAS3 is a member of the protein inhibitor of activated STAT (PIAS) family; however, little is known about the expression and biological functions of PIAS3 in CAC. The aim of our study was to investigate the biological mechanisms of PIAS3 in CAC.
Methods: PIAS3 expression was examined in colon tissues of CAC/CRC patients and azoxymethane-dextran sulfate sodium (AOM-DSS)-induced mice. The role of PIAS3 was studied using a series of in vitro, in vivo and clinical approaches.
Results: Downregulated PIAS3 expression, upregulated miR-18a expression and highly activated NF-κB and STAT3 were observed in colon tissues of CAC/CRC patients and AOM-DSS-induced mice. In vitro experiments revealed that PIAS3 significantly inhibited the activation of NF-κB and STAT3 and demonstrated that activated NF-κB and STAT3 transcriptionally regulated miR-18a level, and up-regulation of miR-18a expression led to defective PIAS3 expression. Moreover, PIAS3-mediated autoregulatory feedback loops (PIAS3/NF-κB/miR-18a and PIAS3/STAT3/miR-18a) were verified in vitro and were found to regulate cell proliferation. Additionally, modulation of the feedback loops via overexpression of PIAS3 or knockdown of miR-18a significantly inhibited cell proliferation in a mouse CRC xenograft model. Furthermore, upregulation of PIAS3 by intracolonic administration of PIAS3 lentivirus or anti-miR-18a lentivirus in AOM-DSS-induced mice led to dramatically reduced tumor sizes/numbers, whereas knockdown of PIAS3 in CAC mice significantly promoted tumor growth.
Conclusion: Our data clearly show that PIAS3-mediated feedback loops control cell proliferation and function as robust driving forces for CAC progression. Targeting these highly activated feedback loops might offer promising therapeutic strategies for CAC.
Keywords: PIAS3, miR-18a, STAT3, NF-κB, colitis-associated colorectal cancer
 Global reach, higher impact
Global reach, higher impact