13.3
Impact Factor
Theranostics 2018; 8(13):3437-3460. doi:10.7150/thno.23853 This issue Cite
Research Paper
Image-guided surgery using near-infrared Turn-ON fluorescent nanoprobes for precise detection of tumor margins
1. Department of Physiology and Pharmacology, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv 69978, Israel.
2. Department of Organic Chemistry, School of Chemistry, Raymond and Beverly Sackler Faculty of Exact Sciences, Tel Aviv University, Tel Aviv 69978, Israel.
3. The Institute for Drug Research, School of Pharmacy, Faculty of Medicine, Campus Ein Kerem, The Hebrew University of Jerusalem, Jerusalem, Israel.
4. Department of Pathology, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv 69978, Israel.
5. Department of Neurosurgery, Tel Aviv Sourasky Medical Center, Israel
* Equal contribution
Abstract
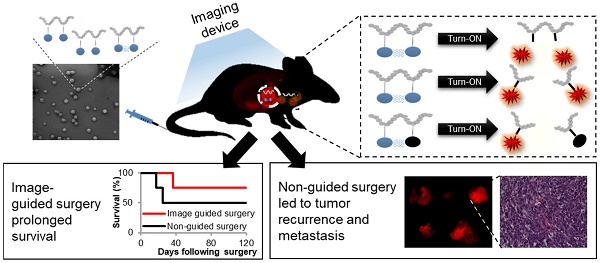
Complete tumor removal during surgery has a great impact on patient survival. To that end, the surgeon should detect the tumor, remove it and validate that there are no residual cancer cells left behind. Residual cells at the incision margin of the tissue removed during surgery are associated with tumor recurrence and poor prognosis for the patient. In order to remove the tumor tissue completely with minimal collateral damage to healthy tissue, there is a need for diagnostic tools that will differentiate between the tumor and its normal surroundings.
Methods: We designed, synthesized and characterized three novel polymeric Turn-ON probes that will be activated at the tumor site by cysteine cathepsins that are highly expressed in multiple tumor types. Utilizing orthotopic breast cancer and melanoma models, which spontaneously metastasize to the brain, we studied the kinetics of our polymeric Turn-ON nano-probes.
Results: To date, numerous low molecular weight cathepsin-sensitive substrates have been reported, however, most of them suffer from rapid clearance and reduced signal shortly after administration. Here, we show an improved tumor-to-background ratio upon activation of our Turn-ON probes by cathepsins. The signal obtained from the tumor was stable and delineated the tumor boundaries during the whole surgical procedure, enabling accurate resection.
Conclusions: Our findings show that the control groups of tumor-bearing mice, which underwent either standard surgery under white light only or under the fluorescence guidance of the commercially-available imaging agents ProSense® 680 or 5-aminolevulinic acid (5-ALA), survived for less time and suffered from tumor recurrence earlier than the group that underwent image-guided surgery (IGS) using our Turn-ON probes. Our "smart" polymeric probes can potentially assist surgeons' decision in real-time during surgery regarding the tumor margins needed to be removed, leading to improved patient outcome.
Keywords: precision nanomedicine, polymers, PGA, HPMA copolymer, theranostics, molecular imaging, NIR fluorescence, image-guided surgery
 Global reach, higher impact
Global reach, higher impact