Impact Factor
Theranostics 2018; 8(14):3824-3840. doi:10.7150/thno.25308 This issue Cite
Research Paper
Targeting S1PR1/STAT3 loop abrogates desmoplasia and chemosensitizes pancreatic cancer to gemcitabine
1. Cancer Research Program, Rajiv Gandhi Centre for Biotechnology, Thiruvananthapuram, Kerala State, India 695014
2. Ph.D scholar, Manipal Academy of Higher Education (MAHE), Manipal, India
3. Division of Molecular and Cellular Medicine, National Cancer Center Research Institute, Tsukiji 5-1-1, Chuo-ku, Tokyo 104-0045, Japan
4. Department of Biostatistics, National Institute of Mental and Neurosciences, Bengaluru, India 560029
5. Department of Pathology, Regional Cancer Centre, Thiruvananthapuram, India 695025
Abstract
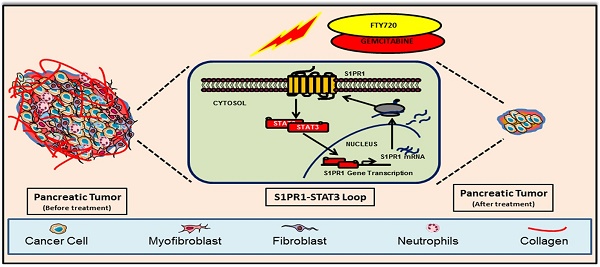
Rationale: Pancreatic cancer is associated with poor prognosis with a 5-year survival rate of less than 6%. Approximately 90% of pancreatic cancer patients harbor somatic mutations in the KRAS gene. Multiple lines of evidence suggest a persistent activation of STAT3 in KRAS-driven oncogenesis contributing to desmoplasia and gemcitabine resistance. Sphingosine 1-phosphate receptor 1 (S1PR1) is an integral component of tumor progression and maintains an activated state of STAT3. FTY720 is an approved drug for multiple sclerosis and acts as a functional antagonist for S1PR1. Here we explored the potential utility of FTY720 to target S1PR1/STAT3 and other major signaling pathways in pancreatic cancer, and sought proof-of-principle for repurposing FTY720 for the treatment of pancreatic cancer.
Methods: We examined the activity of FTY720 in the proliferation, apoptosis, and cell cycle assays in human and mouse pancreatic cancer model systems. Further, we studied the efficacy of using a combination of FTY720 and gemcitabine as opposed to individual agents in vitro as well as in vivo
Results: Treatment of human and mouse pancreatic cancer cells with FTY720 resulted in inhibition of growth, increased apoptosis, and cell cycle arrest. FTY720 in combination with gemcitabine breached the mitochondrial membrane potential, altered the S1PR1-STAT3 loop, and inhibited epithelial to mesenchymal (EMT) transition. Data from murine models exhibited a marked reduction in the tumor size, increased apoptosis, inhibited NF-κB, S1PR1/STAT3, Shh signaling and desmoplasia, modulated the expression of gemcitabine-metabolizing transport enzymes, and restored the expression of tumor suppressor gene PP2A.
Conclusion: Taken together, our results established FTY720 as a propitious molecule, which increases the efficacy of gemcitabine and represents a promising agent in the management of pancreatic cancer.
Keywords: pancreatic cancer, FTY720, S1P receptor 1, PP2A, metastasis, gemcitabine, STAT3
 Global reach, higher impact
Global reach, higher impact