13.3
Impact Factor
Theranostics 2018; 8(15):4226-4237. doi:10.7150/thno.23084 This issue Cite
Research Paper
Nerve-targeted probes for fluorescence-guided intraoperative imaging
1. Department of Surgery, Division of Otolaryngology-Head and Neck Surgery, University of California at San Diego, La Jolla, California, USA
2. Department of Pharmacology, University of California at San Diego, La Jolla, California, USA.
3. Howard Hughes Medical Institute, University of California at San Diego, La Jolla, California, USA
4. Department of Urology, UC San Diego Health System, La Jolla, California, USA.
5. Department of Otorhinolaryngology, Ulsan University Hospital, University of Ulsan College of Medicine, Ulsan, Republic of Korea
*Authors contributed equally
# Author is deceased
Abstract
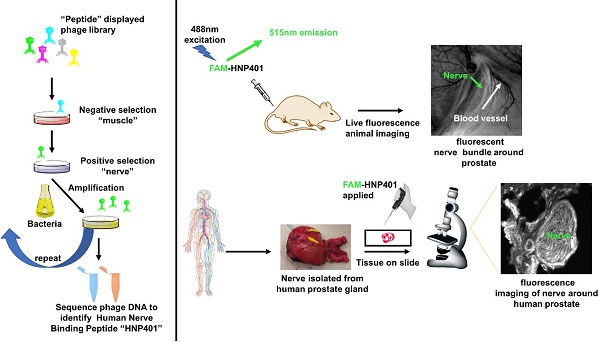
A fundamental goal of many surgeries is nerve preservation, as inadvertent injury can lead to patient morbidity including numbness, pain, localized paralysis and incontinence. Nerve identification during surgery relies on multiple parameters including anatomy, texture, color and relationship to surrounding structures using white light illumination. We propose that fluorescent labeling of nerves can enhance the contrast between nerves and adjacent tissue during surgery which may lead to improved outcomes.
Methods: Nerve binding peptide sequences including HNP401 were identified by phage display using selective binding to dissected nerve tissue. Peptide dye conjugates including FAM-HNP401 and structural variants were synthesized and screened for nerve binding after topical application on fresh rodent and human tissue and in-vivo after systemic IV administration into both mice and rats. Nerve to muscle contrast was quantified by measuring fluorescent intensity after topical or systemic administration of peptide dye conjugate.
Results: Peptide dye conjugate FAM-HNP401 showed selective binding to human sural nerve with 10.9x fluorescence signal intensity (1374.44 ± 425.96) compared to a previously identified peptide FAM-NP41 (126.17 ± 61.03). FAM-HNP401 showed nerve-to-muscle contrast of 3.03 ± 0.57. FAM-HNP401 binds and highlight multiple human peripheral nerves including lower leg sural, upper arm medial antebrachial as well as autonomic nerves isolated from human prostate.
Conclusion: Phage display has identified a novel peptide that selectively binds to ex-vivo human nerves and in-vivo using rodent models. FAM-HNP401 or an optimized variant could be translated for use in a clinical setting for intraoperative identification of human nerves to improve visualization and potentially decrease the incidence of intra-surgical nerve injury.
Keywords: human nerve, fluorescence imaging, nerve targeting, translational
 Global reach, higher impact
Global reach, higher impact