Impact Factor
Theranostics 2018; 8(17):4695-4709. doi:10.7150/thno.26203 This issue Cite
Research Paper
Gαi1 and Gαi3mediate VEGF-induced VEGFR2 endocytosis, signaling and angiogenesis
1. Department of Anesthesiology, Huai'an Maternity and Child Healthcare Hospital, Yangzhou University Medical School, Huai'an, China
2. The Fourth School of Clinical Medicine, The Affiliated Eye Hospital, Nanjing Medical University, Nanjing 210029, China
3. Department of Vascular Surgery, Renji Hospital, School of Medicine, Shanghai Jiaotong University, Shanghai, China. Pujian Road 160, Shanghai, China
4. Jiangsu Key Laboratory of Neuropsychiatric Diseases and Institute of Neuroscience, Soochow University, Suzhou, China.
5. North District, The Municipal Hospital of Suzhou, Suzhou, China
# Co-first authors.
Abstract
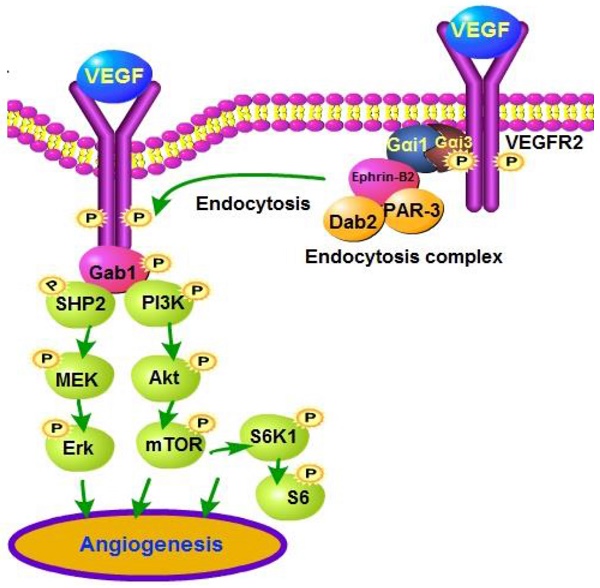
VEGF binding to VEGFR2 leads to VEGFR2 endocytosis and downstream signaling activation to promote angiogenesis.
Methods: Using genetic strategies, we tested the requirement of α subunits of heterotrimeric G proteins (Gαi1/3) in the process.
Results: Gαi1/3 are located in the VEGFR2 endocytosis complex (VEGFR2-Ephrin-B2-Dab2-PAR-3), where they are required for VEGFR2 endocytosis and downstream signaling transduction. Gαi1/3 knockdown, knockout or dominant negative mutation inhibited VEGF-induced VEGFR2 endocytosis, and downstream Akt-mTOR and Erk-MAPK activation. Functional studies show that Gαi1/3 shRNA inhibited VEGF-induced proliferation, invasion, migration and vessel-like tube formation of HUVECs. In vivo, Gαi1/3 shRNA lentivirus inhibited alkali burn-induced neovascularization in mouse cornea. Further, oxygen-induced retinopathy (OIR)-induced retinal neovascularization was inhibited by intravitreal injection of Gαi1/3 shRNA lentivirus. Moreover, in vivo angiogenesis by alkali burn and OIR was significantly attenuated in Gαi1/3 double knockout mice. Significantly, Gαi1/3 proteins are upregulated in proliferative retinal tissues of proliferative diabetic retinopathy (PDR) patients.
Conclusion: These results provide mechanistic insights into the critical role played by Gαi1/3 proteins in VEGF-induced VEGFR2 endocytosis, signaling and angiogenesis.
Keywords: VEGFR2, Gαi1, Gαi3, endocytosis, angiogenesis, signaling
 Global reach, higher impact
Global reach, higher impact