Impact Factor
Theranostics 2018; 8(17):4781-4794. doi:10.7150/thno.25189 This issue Cite
Research Paper
RGS4 deficit in prefrontal cortex contributes to the behaviors related to schizophrenia via system xc--mediated glutamatergic dysfunction in mice
1. School of Medicine, China Medical University, Taichung, Taiwan
2. Department of Psychiatry, Chiayi Branch, Taichung Veterans General Hospital, Chiayi, Taiwan
3. Graduate Institute of Biomedical Science, China Medical University, Taichung, Taiwan
4. National PET/Cyclotron Center and Department of Nuclear Medicine, Taipei Veterans General Hospital, Taipei, Taiwan
5. Department of Neurology, Center for Translational Medicine, and Graduate Institute of Biomedical Science, China Medical University and Hospital, Taichung, Taiwan
6. Department of Medical Research, China Medical University Hospital, Taichung, Taiwan
7. Department of Biomedical Informatics, Asia University, Taichung, Taiwan
* these authors contributed equally to this work
Abstract
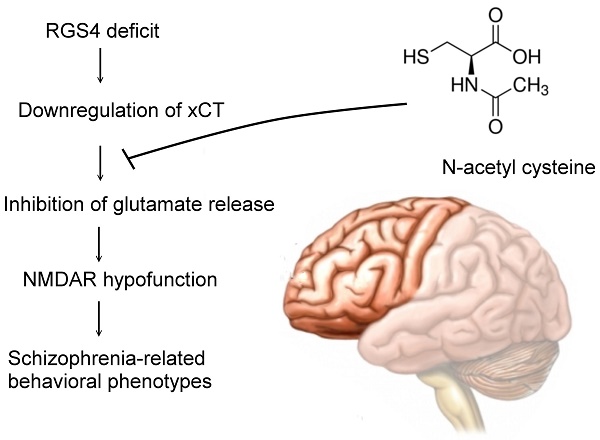
Rationale: Although molecular investigations of regulator of G-protein signaling 4 (RGS4) alterations in schizophrenia patients yielded partially inconsistent findings, the previous studies suggested that RGS4 is both a positional and functional candidate gene for schizophrenia and is significantly decreased in the prefrontal cortex. However, the exact role of RGS4 in the pathophysiology of schizophrenia is unclear. Moreover, a whole genome transcription profile study showed the possibility of RGS4-regulated expression of SLC7A11(xCT), a component of cysteine/glutamate transporter or system xc-. We hypothesized that system xc- is a therapeutic target of RGS4 deficit-mediated schizophrenia.
Methods: Pharmacological and genetic manipulation of RGS4 in organotypic brain slice cultures were used as an ex vivo model to investigate its role in system xc- and glutamatergic function. Lentiviral-based mouse models with RGS4 deficit in the prefrontal cortex and treatment with system xc- activator, N-acetyl cysteine (NAC), were utilized to observe their impacts on glutamatergic function and schizophrenic behaviors.
Results: Genetic and pharmacological inhibition of RGS4 resulted in a significant decrease in SLC7A11 (xCT) expression and hypofunction of system xc- and reduced glutamatergic function in organotypic brain slice cultures. However, NAC restored the dysregulation of RGS4-mediated functional deficits of glutamate. Moreover, knockdown of RGS4 specifically in the prefrontal cortex caused mice to exhibit behaviors related to schizophrenia such as increased stereotypy, impaired prepulse inhibition, deficits in social interactions, working memory, and nesting behavior, while enhancing sensitivity to the locomotor stimulatory effect of MK-801. These mice displayed glutamatergic dysfunction in the prefrontal cortex, which may have contributed to the behavioral deficits. RGS4 knockdown mice that received NAC treatment had improved glutamatergic dysfunction and schizophrenia behaviors.
Conclusion: Our results suggest that RGS4 deficit induces dysregulation and dysfunction of system xc-, which further results in functional deficits of the glutamatergic system and subsequently to schizophrenia-related behavioral phenotypes. Activation of system xc- offers a promising strategy to treat RGS4 deficit-mediated schizophrenia.
Keywords: regulator of G-protein signaling 4, system xc-, schizophrenia, glutamatergic transmission, N-acetyl cysteine
 Global reach, higher impact
Global reach, higher impact