13.3
Impact Factor
Theranostics 2019; 9(1):104-125. doi:10.7150/thno.30308 This issue Cite
Review
Selective Autophagy Regulates Cell Cycle in Cancer Therapy
1. School of Pharmaceutical Sciences, Health Science Center, Shenzhen University, Shenzhen, 518060, PR China;
2. Division of Molecular Pharmacology of Infectious Agents, Department of Molecular Microbiology and Immunology, Nagasaki University, Nagasaki, 852-8521, Japan
3. College of Life Science and Technology, Guangzhou Jinan Biomedicine Research and Development Center, Jinan University, Guangzhou 510632, PR China
Abstract
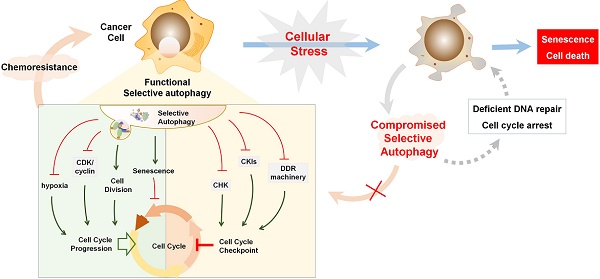
Aberrant function of cell cycle regulators results in uncontrolled cell proliferation, making them attractive therapeutic targets in cancer treatment. Indeed, survival of many cancers exclusively relies on these proteins, and several specific inhibitors are in clinical use. Although the ubiquitin-proteasome system is responsible for the periodic quality control of cell cycle proteins during cell cycle progression, increasing evidence clearly demonstrates the intimate interaction between cell cycle regulation and selective autophagy, important homeostasis maintenance machinery. However, these studies have often led to divergent rather than unifying explanations due to complexity of the autophagy signaling network, the inconsistent functions between general autophagy and selective autophagy, and the different characteristics of autophagic substrates. In this review, we highlight current data illustrating the contradictory and important role of cell cycle proteins in regulating autophagy. We also focus on how selective autophagy acts as a central mechanism to maintain orderly DNA repair and genome integrity by degrading specific cell cycle proteins, regulating cell division, and promoting DNA damage repair. We further discuss the ways in which selective autophagy may impact the cell cycle regulators, since failure to appropriately remove these can interfere with cell death-related processes, including senescence and autophagy-related cell death. Imbalanced cell proliferation is typically utilized by cancer cells to acquire resistance. Finally, we discuss the possibility of a potent anticancer therapeutic strategy that targets selective autophagy or autophagy and cell cycle together.
Keywords: selective autophagy, chemoresistance, senescence, DNA damage repair, cell cycle checkpoints
 Global reach, higher impact
Global reach, higher impact