13.3
Impact Factor
Theranostics 2019; 9(1):152-166. doi:10.7150/thno.27175 This issue Cite
Research Paper
Multimodal and Multiscale Analysis Reveals Distinct Vascular, Metabolic and Inflammatory Components of the Tissue Response to Limb Ischemia
1. Vascular Medicine Research, Dept. of Nephrology and Hypertension, Hannover Medical School, Hannover, Germany
2. Dept. of Nephrology and Hypertension, Hannover Medical School, Hannover, Germany
3. Dept. of Nuclear Medicine, Hannover Medical School, Hannover, Germany
4. Dept. of Diagnostic and Interventional Radiology, Hannover Medical School, Hannover, Germany
5. Central Research Facility Cell Sorting, Hannover Medical School, Hannover, Germany,
6. Dept. of Plastic, Aesthetic, Reconstructive Surgery, Hannover Medical School, Hannover, Germany
* equal contribution
Abstract
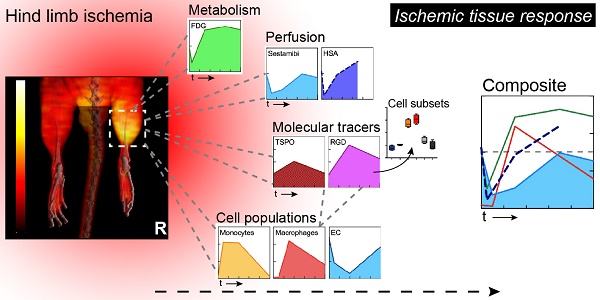
Ischemia triggers a complex tissue response involving vascular, metabolic and inflammatory changes.
Methods: We combined hybrid SPECT/CT or PET/CT nuclear imaging studies of perfusion, metabolism and inflammation with multicolor flow cytometry-based cell population analysis to comprehensively analyze the ischemic tissue response and to elucidate the cellular substrate of noninvasive molecular imaging techniques in a mouse model of hind limb ischemia.
Results: Comparative analysis of tissue perfusion with [99mTc]-Sestamibi and arterial influx with [99mTc]-labeled albumin microspheres by SPECT/CT revealed a distinct pattern of response to vascular occlusion: an early ischemic period of matched suppression of tissue perfusion and arterial influx, a subacute ischemic period of normalized arterial influx but impaired tissue perfusion, and a protracted post-ischemic period of hyperdynamic arterial and normalized tissue perfusion, indicating coordination of macrovascular and microvascular responses. In addition, the subacute period showed increased glucose uptake by [18F]-FDG PET/CT scanning as the metabolic response of viable tissue to hypoperfusion. This was associated with robust macrophage infiltration by flow cytometry, and glucose uptake studies identified macrophages as major contributors to glucose utilization in ischemic tissue. Furthermore, imaging with the TSPO ligand [18F]-GE180 showed a peaked response during the subacute phase due to preferential labeling of monocytes and macrophages, while imaging with [68Ga]-RGD, an integrin ligand, showed prolonged post-ischemic upregulation, which was attributed to labeling of macrophages and endothelial cells by flow cytometry.
Conclusion: Combined nuclear imaging and cell population analysis reveals distinct components of the ischemic tissue response and associated cell subsets, which could be targeted for therapeutic interventions.
Keywords: ischemia, vascular, metabolic, inflammatory
 Global reach, higher impact
Global reach, higher impact