Impact Factor
Theranostics 2019; 9(1):167-178. doi:10.7150/thno.28033 This issue Cite
Research Paper
Enhancement of mitochondrial ROS accumulation and radiotherapeutic efficacy using a Gd-doped titania nanosensitizer
1. College of Chemistry, Chemical Engineering and Materials Science, Key Laboratory of Molecular and Nano Probes, Ministry of Education, Collaborative Innovation Center of Functionalized Probes for Chemical Imaging in Universities of Shandong, Institute of Molecular and Nano Science, Shandong Normal University, Jinan 250014, P. R. China.
2. Radiation Department, Qilu Hospital of Shandong University, Jinan 250100, P. R. China.
*These authors contributed equally to this work.
Abstract
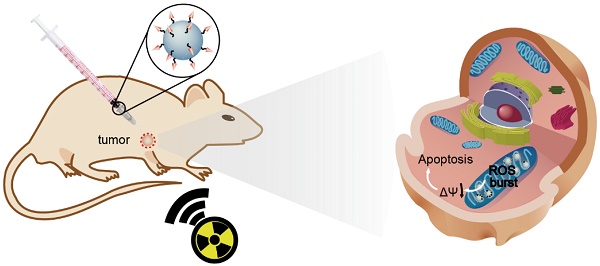
Radiotherapy is an extensively used treatment modality in the clinic and can kill malignant cells by generating cytotoxic reactive oxygen species (ROS). Unfortunately, excessive dosages of radiation are typically required because only a small proportion of the radiative energy is adsorbed by the soft tissues of a tumor, which results in the nonselective killing of normal cells and severe systemic side effects. An efficient nanosensitizer that makes cancer cells more sensitive to radiotherapy under a relatively low radiation dose would be highly desirable.
Methods: In this study, we developed a Gd-doped titania nanosensitizer that targets mitochondria to achieve efficient radiotherapy. Upon X-ray irradiation, the nanosensitizer triggers a “domino effect” of ROS accumulation in mitochondria. This overabundance of ROS leads to mitochondrial permeability transition and ultimately irreversible cell apoptosis. Confocal laser imaging, western blotting and flow cytometry analysis were used to explore the biological process of intrinsic apoptosis induced by the nanosensitizer. Clonogenic survival assay, cell migration and invasion experiments were employed to evaluate the radiosensitizing effect of the nanosensitizer in vitro. Finally, to evaluate the therapeutic outcome of the nanosensitizer in vivo, MCF-7 tumor model was used.
Results: Confocal laser images and western blotting data demonstrated that the nanosensitizer in conjunction with X-ray irradiation could induce cell apoptosis in ROS-mediated apoptotic signal pathways. A clonogenic survival assay revealed that cells treated with the prepared nanosensitizer exhibited a lower number of viable cell colonies than that of the nontargeted group under X-ray irradiation. Notably, with only a single dose of radiotherapy, the mitochondria-targeted nanosensitizer elicited the complete ablation of tumors in a mouse model.
Conclusion: The designed nanosensitizer in combination with X-ray radiation exposure could be used for radiotherapy against cancer in living cells and in vivo. Moreover, the nanosensitizer with mitochondria targeting played a pivotal role in triggering a “domino effect” of ROS and cell apoptosis. The current strategy could provide new opportunities in designing efficient radiosensitizers for future cancer therapy.
Keywords: nanosensitizer, mitochondria-targeting, reactive oxygen species, radiotherapy, cancer
 Global reach, higher impact
Global reach, higher impact