Impact Factor
Theranostics 2019; 9(1):246-264. doi:10.7150/thno.30174 This issue Cite
Research Paper
Near-infrared light-regulated cancer theranostic nanoplatform based on aggregation-induced emission luminogen encapsulated upconversion nanoparticles
1. The Key Laboratory of Biomedical Information Engineering of Ministry of Education, School of Life Science and Technology, Xi'an Jiaotong University, Shaanxi, 710049, China
2. Bioinspired Engineering & Biomechanics Center (BEBC), Xi'an Jiaotong University, Shaanxi, 710049, China
3. State Key Laboratory for Mechanical Behavior of Materials, School of Materials Science and Engineering, Xi'an Jiaotong University, Xi'an 710049, People's Republic of China
4. Department of Biomedical Engineering, Southern University of Science and Technology, Shenzhen, Guangdong 510855, China.
5. Department of Chemistry, Institute for Advanced Study, Institute of Molecular Functional Materials, Division of Biomedical Engineering, Division of Life Science, and State Key Laboratory of Molecular Neuroscience, The Hong Kong University of Science & Technology, Clear Water Bay, Kowloon, Hong Kong, China
6. Guangdong Innovative Research Team, SCUT-HKUST Joint Research Laboratory, State Key Laboratory of Luminescent Materials and Devices, South China University of Technology, Guangzhou, 510640, China
7. Department of Chemical and Biomolecular Engineering, National University of Singapore, 4 Engineering Drive 4, 117585, Singapore.
*Authors contributed equally
Abstract
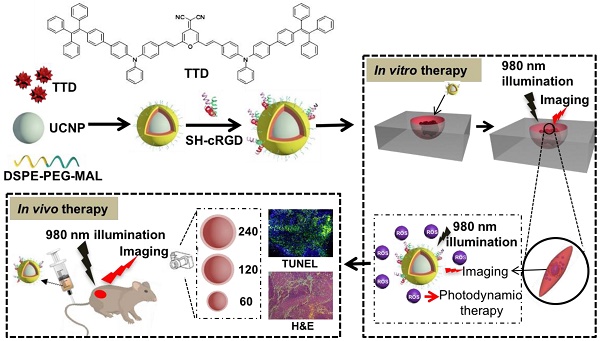
Photodynamic therapy (PDT) has been widely applied in the clinic for the treatment of various types of cancer due to its precise controllability, minimally invasive approach and high spatiotemporal accuracy as compared with conventional chemotherapy. However, the porphyrin-based photosensitizers (PSs) used in clinics generally suffer from aggregation-caused reductions in the generation of reactive oxygen species (ROS) and limited tissue penetration because of visible light activation, which greatly hampers their applications for the treatment of deep-seated tumors.
Methods: We present a facile strategy for constructing a NIR-regulated cancer theranostic nanoplatform by encapsulating upconversion nanoparticles (UCNPs) and a luminogen (2-(2,6-bis((E)-4-(phenyl(40-(1,2,2-triphenylvinyl)-[1,10-biphenyl]-4-yl)amino)styryl)-4H-pyran-4-ylidene)malononitrile, TTD) with aggregation-induced emission (AIEgen) characteristics using an amphiphilic polymer, and further conjugating cyclic arginine-glycine-aspartic acid (cRGD) peptide to yield UCNP@TTD-cRGD NPs. We then evaluated the bioimaging and anti-tumor capability of the UCNP@TTD-cRGD NPs under NIR light illumination in an in vitro three-dimensional (3D) cancer spheroid and in a murine tumor model, respectively.
Results: With a close match between the UCNP emission and absorption of the AIEgen, the synthesized NPs could efficiently generate ROS, even under excitation through thick tissues. The NIR-regulated UCNP@TTD-cRGD NPs that were developed could selectively light up the targeted cancer cells and significantly inhibit tumor growth during the NIR-regulated PDT treatment as compared with the cells under white light excitation.
Conclusion: In summary, the synthesized UCNP@TTD-cRGD NPs showed great potential in NIR light-regulated photodynamic therapy of deep-seated tumors. Our study will inspire further exploration of novel theranostic nanoplatforms that combine UCNPs and various AIEgen PSs for the advancement of deep-seated tumor treatments with potential clinical translations.
Keywords: aggregation-induced emission (AIE), photodynamic therapy, near infrared light, active targeting, tumor imaging
 Global reach, higher impact
Global reach, higher impact