13.3
Impact Factor
Theranostics 2019; 9(8):2198-2208. doi:10.7150/thno.30798 This issue Cite
Research Paper
Long noncoding RNA TUG1 inhibits osteogenesis of bone marrow mesenchymal stem cells via Smad5 after irradiation
Department of Blood Transfusion, Lab of Radiation biology, The Second Affiliated Hospital, Third Military Medical University, Chongqing, 400037, China.
*These authors contributed equally to this work and should be considered co-first authors.
Abstract
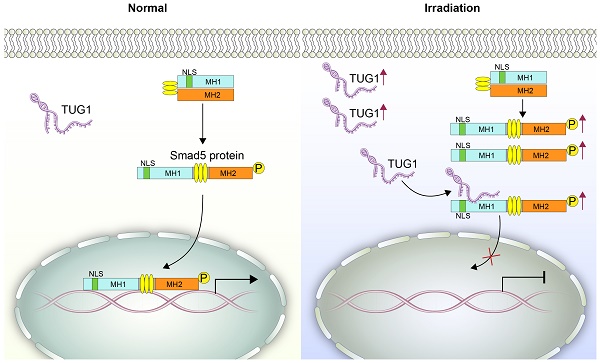
Irradiation can greatly inhibit osteogenesis of bone marrow mesenchymal stem cells (BM-MSCs). However, the mechanism remains unclear.
Methods: We analyzed the expression profile of long noncoding RNAs (lncRNAs) in BM-MSCs using microarray data. LncRNA TUG1 (Taurine Upregulated Gene 1) was selected and tested in radiated BM-MSCs and non-radiated BM-MSCs. Functional analyses (in vitro) were performed to confirm the role of TUG1 in the osteogenic inhibition induced by irradiation. A RIP (RNA immunoprecipitation) assay was performed to detect the interaction of TUG1 and Smad5. Smad5 and the phosphorylated Smad5 (p-Smad5) were tested by western blot. The nuclear translocation of p-Smad5 were tested by immunofluorescence analysis. Furthermore, a series of Smad5 deletions was constructed to identify the TUG1 binding site of Smad5.
Results: We found that numerous lncRNAs, including TUG1, exhibit significant expression differences after irradiation. After irradiation TUG1 was significantly increased in BM-MSCs and inhibited osteogenesis. Furthermore, TUG1 directly bound to Smad5, an osteogenic enhancer. Although the phosphorylation level of Smad5 was increased following irradiation, osteogenesis of BM-MSCs was decreased. Mechanistically, TUG1 interacting with the 50-90 aa region of Smad5 and blocks the nuclear translocation of p-Smad5, abolishing osteogenic signalling after irradiation.
Conclusion: These results indicate that TUG1 is a negative regulator of Smad5 signalling and suppresses osteogenesis of BM-MSCs after irradiation.
Keywords: TUG1, Osteogenesis, Bone Marrow Mesenchymal Stem Cells, Smad5, Irradiation
 Global reach, higher impact
Global reach, higher impact