13.3
Impact Factor
Theranostics 2019; 9(12):3515-3525. doi:10.7150/thno.32742 This issue Cite
Research Paper
GSH Activated Biotin-tagged Near-Infrared Probe for Efficient Cancer Imaging
1. College of Pharmaceutical Sciences, Zhejiang University, Hangzhou 310058, China
2. Translational Medicine Research Center, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine, Hangzhou 310006, China
3. Department of Pathology, The Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou 310009, China
*These authors contributed equally to this work.
Abstract
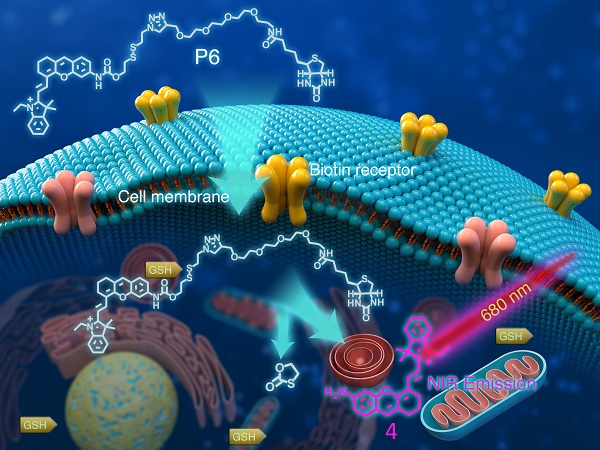
Tumor imaging tools with high specificity and sensitivity are needed to aid the boundary recognition in solid tumor diagnosis and surgical resection. In this study, we developed a near infra-red (NIR) probe (P6) for in vitro/in vivo tumor imaging on the basis of the dual strategy of cancer cell targeting and stimulus-dependent activation. The selective imaging capacity towards cancer cells of P6 was thoroughly investigated, and the potential mechanisms of endocytosis were preliminary explored.
Methods: GSH-activated biotin labelled NIR probe (P6) was designed, synthesized and characterized. The GSH responsive properties were systematically illustrated through UV-vis, fluorescent tests and LC-MS analysis. In vitro fluorescent imaging of probe P6 was collected in various living cancer cell lines (i.e. SW480, HGC-27, H460, BxPC-3, KHOS) and normal cell lines (i.e. BEAS-2B, HLF-1, THP1) under confocal laser scanning microscopy. Probe P6 was further applied to image primary human cancer cells which were freshly isolated from the peritoneal carcinoma and rectal cancer patients. Serial sections of human tumor tissues were collected and sent for H&E (hematoxylin-eosin) staining and P6 imaging. Live fluorescent and photoacoustic imaging were used to investigate the in vivo imaging of P6 in both tumor and normal tissues in HGC-27 and KHOS xenograft model.
Results: Probe P6 could be recognized and transported into cancer cells by tumor specific biotin receptors and efficiently be triggered by GSH to release fluorophore 4. In fact, the cellular uptake of P6 could be partially blocked by the addition of free biotin. Furthermore, probe P6 could image various cancer cell lines, as well as primary cancer cells, exhibiting a ten-fold increase in fluorescence intensity over normal cells. In freshly dissected cancer tissues, P6 fluorescent imaging distinguished the cancerous area under confocal laser scanning microscopy, which was exact the same area as indicated by H&E staining. We also found that P6 exhibited superior selectivity against cancer tissues by local injection.
Conclusion: In this study, we developed a dual-modal NIR probe P6 with enhanced cellular uptake into cancer cells and environmental stimulus triggered fluorescence. Our strategy provided a novel insight into the development of imaging tools that could be potentially used for fluorescent image-guided cancer boundary recognition and possibly cancer diagnosis.
Keywords: GSH activation, Near-Infrared (NIR) imaging, boundary recognition, In Vivo Imaging, histopathological analyses
 Global reach, higher impact
Global reach, higher impact