13.3
Impact Factor
Theranostics 2019; 9(13):3732-3753. doi:10.7150/thno.34377 This issue Cite
Research Paper
A Multifunctional Nanotherapy for Targeted Treatment of Colon Cancer by Simultaneously Regulating Tumor Microenvironment
1. Department of Pharmaceutics, College of Pharmacy, Third Military Medical University, Chongqing 400038, China
2. Department of Pharmaceutical Engineering, College of Pharmaceutical Sciences, Southwest University, Chongqing 400715, China
3. Department of Neurology, Southwest Hospital, Third Military Medical University, Chongqing 400038, China
* These authors contributed equally to this work.
Abstract
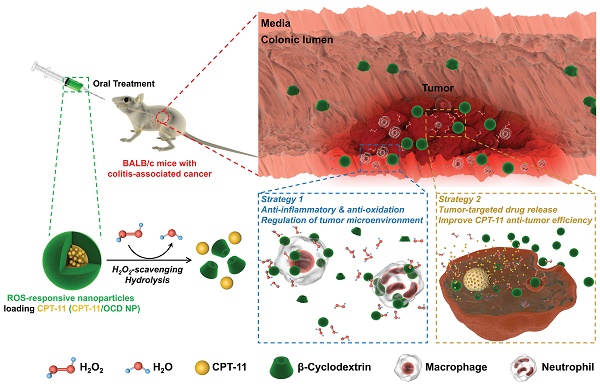
Colitis-associated colon cancer (CAC) is a widely recognized cancer, while treatment with the existing chemotherapeutic drugs affords limited clinical benefits. Herein we proposed a site-specific, combination nanotherapy strategy for targeted treatment of CAC by the oral route.
Methods: A reactive oxygen species (ROS)-responsive and hydrogen peroxide-eliminating material OCD was synthesized, which was further produced into a functional nanoparticle (OCD NP). The antioxidative stress and anti-inflammatory effects of OCD NP were examined by in vitro and in vivo experiments. By packaging an anticancer drug camptothecin-11 (CPT-11) into OCD NP, a ROS-responsive nanotherapy CPT-11/OCD NP was obtained, and its antitumor activity was evaluated by both in vitro and in vivo studies. Preliminary safety studies were also performed for CPT-11/OCD NP in mice.
Results: OCD NP significantly attenuated oxidative stress and inhibited inflammatory response in different cells and mice with induced colitis. CPT-11/OCD NP could selectively release drug molecules under intestinal pH conditions and at high levels of ROS. In C26 murine colon carcinoma cells, this nanotherapy showed significantly higher antitumor activity compared to free CPT-11 and a non-responsive CPT-11 nanotherapy. Correspondingly, oral delivery of CPT-11/OCD NP notably inhibited tumorigenesis and tumor growth in mice with induced CAC. By combination therapy with the nanovehicle OCD NP in the inflammatory phase, more desirable therapeutic effects were achieved. Furthermore, CPT-11/OCD NP displayed excellent safety profile for oral administration at a dose that is 87.3-fold higher than that employed in therapeutic studies.
Conclusions: Anticancer nanotherapies derived from intrinsic anti-inflammatory nanocarriers are promising for targeted combination treatment of inflammation-associated tumors by simultaneously shaping pro-inflammatory microenvironment toward a relatively normal niche sensitive to chemotherapy.
Keywords: colitis-associated colon cancer, anti-inflammation, reactive oxygen species, nanoparticle, camptothecin-11, site-specific therapy
 Global reach, higher impact
Global reach, higher impact