Impact Factor
Theranostics 2019; 9(13):3940-3951. doi:10.7150/thno.31647 This issue Cite
Research Paper
Clathrin-mediated Endocytosis of Alpha-1 Antitrypsin is Essential for its Protective Function in Islet Cell Survival
1. Department of Surgery, Medical University of South Carolina, Charleston, 29425
2. Department of Medicine, Medical University of South Carolina, Charleston, 29425
3. Department of Regenerative Medicine and Cell Biology, Medical University of South Carolina, Charleston, 29425
*JW and WG contributed equally and should be considered as co-first authors.
#CS and HW share senior authorship.
Abstract
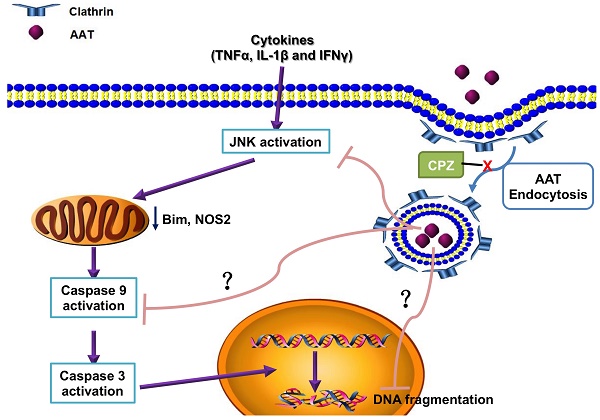
Cytokine-induced pancreatic β cell death plays a pivotal role in both type 1 and type 2 diabetes. Our previous study showed that alpha-1 antitrypsin (AAT) inhibits β cell death through the suppression of cytokine-induced c-Jun N-terminal kinase (JNK) activation in an islet transplantation model. The aim of this study was to further understand how AAT impacts β cells by studying AAT endocytosis in human islets and a βTC3 murine insulinoma cell line.
Methods: In vitro, human islets and βTC3 cells were stimulated with cytokines in the presence or absence of chlorpromazine (CPZ), a drug that disrupts clathrin-mediated endocytosis. Western blot, real-time PCR and cell death ELISA were performed to investigate β cell death. The oxygen consumption rate (OCR) was measured on human islets. In vivo, islets were harvested from C57BL/6 donor mice treated with saline or human AAT and transplanted into the livers of syngeneic mice that had been rendered diabetic by streptozotocin (STZ). Islet graft survival and function were analyzed.
Results: AAT was internalized by β cells in a time- and dose-dependent manner. AAT internalization was mediated by clathrin as treatment with CPZ, profoundly decreased AAT internalization, cytokine-induced JNK activation and the downstream upregulation of c-Jun mRNA expression. Similarly, addition of CPZ attenuated cytokine-induced caspase 9 cleavage (c-casp 9) and DNA fragmentation, which was suppressed by AAT. Treatment of donor mice with AAT produced AAT internalization in islets, and resulted in a higher percentage of recipients reaching normoglycemia after syngeneic intraportal islet transplantation.
Conclusion: Our results suggest that AAT is internalized by β cells through clathrin-mediated endocytosis that leads to the suppression of caspase 9 activation. This process is required for the protective function of AAT in islets when challenged with proinflammatory cytokines or after islet transplantation.
Keywords: Alpha-1 antitrypsin, clathrin-mediated endocytosis, islet transplantation, graft survival, donor treatment
 Global reach, higher impact
Global reach, higher impact