13.3
Impact Factor
Theranostics 2019; 9(14):4006-4018. doi:10.7150/thno.32720 This issue Cite
Research Paper
Alarmin augments the antitumor immunity of lentiviral vaccine in ectopic, orthotopic and autochthonous hepatocellular carcinoma mice
1. Department of Cell Biology, Key Laboratory of Immune Microenvironment and Disease (Ministry of Education), Tianjin Medical University, Qixiangtai Road, Heping District, Tianjin, 300070, China
2. School of Medical Laboratory, Tianjin Medical University, Guangdong Road, Tianjin, 300203, China
* These authors contributed equally as joint first authors.
Abstract
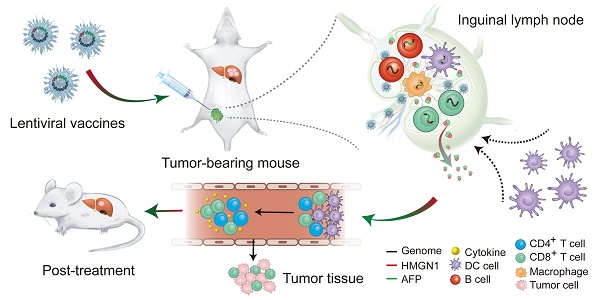
It is a daunting therapeutic challenge to completely eradicate hepatocellular carcinoma (HCC) from patients. Alpha-fetoprotein (AFP) -based vaccines appear promising, however the efficacy needs to be improved.
Methods: Here, we explore if fusing high-mobility group nucleosome binding protein 1 (HMGN1), a potent immunoadjuvant, to AFP (lenti-HA) can augment the antitumor immunity of AFP-expressing lentiviral vector (lenti-AFP), a vehicle extensively employed for genetic immunization with high transduction efficacy and good safety profiles. The antitumor immunity of Lenti-HA was systemically assessed in ectopic, orthotopic and autochthonous HCC models.
Results: Lenti-HA elicited strong anti-HCC effects in mice and amplified the antitumor immunity of lenti-AFP by reducing effective dose 6-fold. Importantly, lenti-HA induced a robust antitumor immune response with prolonged survival rate and improved the immune and tumor microenvironment in mice with carcinogen-induced autochthonous HCC. Lenti-HA localized primarily to lymphoid organs with no preference for specific immune cell types. Activated dendritic cells (DCs), particularly CD103+CD11b- DCs, were also actively recruited to lymph nodes in lenti-HA-treated HCC mice. Moreover, lenti-HA-transduced human DCs elicited stronger immune response than lenti-AFP against HCC cells in vitro.
Conclusion: Our study demonstrates that HMGN1 augments the antitumor immunity of AFP-expressing lentiviral vaccines in HCC mice and human cells in vitro and thus provides a new therapeutic strategy for HCC.
Keywords: HMGN1, lentivirus, immunotherapy, hepatocellular carcinoma
 Global reach, higher impact
Global reach, higher impact