Impact Factor
Theranostics 2019; 9(14):4208-4220. doi:10.7150/thno.33803 This issue Cite
Research Paper
Ubiquitin specific peptidase 5 regulates colorectal cancer cell growth by stabilizing Tu translation elongation factor
1. Suzhou Institute of Systems Medicine, Center for Systems Medicine, Chinese Academy of Medical Sciences, Suzhou, Jiangsu 215123, P. R. China.
2. Department of Pharmacy, The 900th Hospital of the Joint Logistics Support Force, Fuzhou, Fujian 350025, P. R. China
3. Xinhua Hospital, Shanghai Jiaotong University School of Medicine, Shanghai 200092, P. R. China
4. The Asclepius Technology Company Group and Asclepius Cancer Research Center, Suzhou, Jiangsu 215123, P. R. China
5. State Key Laboratory of Innovative Natural Medicine and TCM Injections, Jiangxi Qingfeng pharmaceutical, Ganzhou, Jiangxi 341000, P. R. China
Abstract
Ubiquitin specific peptidase 5 (USP5) is a ubiquitous expressed deubiquitinating enzyme (DUB). It has been shown involved in DNA repair, apoptosis, inflammation, and tumor cell growth. However, the function and molecular mechanism of USP5 in colorectal cancer (CRC) are still unclear. In the present study, we asked how it affected the growth of colorectal cancer cells.
Methods: A shRNA-based high-content screening was performed to identify DUBs affecting the growth of CRC cells. CCK-8 assay and xenografts were used to assess CRC cell growth, survival and tumorigenesis. RT-qPCR, immunoblotting and immunohistochemistry were carried out to quantitate USP5 expression in CRC tissues and cell lines. Immunoprecipitation and mass spectrometry analysis were performed to identify USP5-interacting proteins. Cycloheximide chase was performed to assess Tu translation elongation factor (TUFM) stability. Dual luciferase reporter assay was utilized for USP5 promoter analysis.
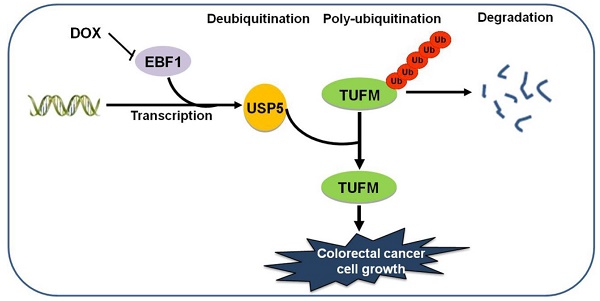
Results: We found that USP5 was highly expressed in a group of primary CRC tissues, and the increased USP5 was correlated with clinical stages and shorter overall survival. While USP5 knockdown effectively inhibited CRC cell growth, overexpressed USP5 promoted the growth of CRC cells and made them more resistant to doxorubicin (DOX). TUFM was discovered as a substrate of USP5. USP5 deubiquitinated TUFM and increased its level in CRC cells. Enforced expression of TUFM was able to alleviate the growth inhibition induced by USP5 knockdown. Further analyses showed that EBF transcription factor 1 (EBF1) was a major regulator for USP5 transcription, and DOX inhibited EBF1-USP5-TUFM axis in CRC cells.
Conclusions: USP5 was required for CRC cells and promoted their growth and resistance to chemotherapeutics. TUFM was a USP5 deubiquitinating substrate that mediated the cellular effects of USP5. The transcription of USP5 was regulated by EBF1. Thus, targeting EBF1-USP5-TUFM axis is a potential novel strategy for CRC treatment.
Keywords: Deubiquitinase, USP5, Colorectal cancer, TUFM, EBF1
 Global reach, higher impact
Global reach, higher impact