13.3
Impact Factor
Theranostics 2019; 9(15):4437-4449. doi:10.7150/thno.34953 This issue Cite
Research Paper
Novel ginsenoside-based multifunctional liposomal delivery system for combination therapy of gastric cancer
1. Department of Pharmaceutics, School of Pharmacy, Fudan University & Key Laboratory of Smart Drug Delivery, Ministry of Education, Shanghai 201203, China
2. Shanghai Ginposome Pharmatech Co., Ltd, Shanghai 201600, China
3. Institute of Tropical Medicine, Guangzhou University of Chinese Medicine, Guangzhou 510006, China
4. Institute of Clinical Pharmacology, Guangzhou University of Traditional Chinese Medicine, Guangzhou 510006, China
5. Institute of Integrated Chinese and Western Medicine, Fudan University, Shanghai 200040, China
#Chao Hong and Dan Wang contributed equally to this work
Abstract
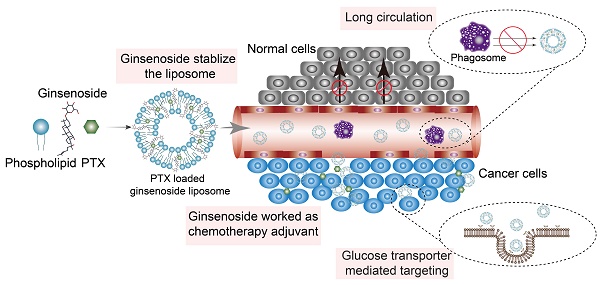
The clinical treatment of gastric cancer (GC) is hampered by the development of anticancer drug resistance and the unfavorable pharmacokinetics, off-target toxicity, and inadequate intratumoral accumulation of the current chemotherapy treatments. Ginsenosides combined with paclitaxel (PTX) have been shown to exert synergistic inhibition of human GC cell proliferation. In the present study, we developed a novel multifunctional liposome system, in which ginsenosides functioned as the chemotherapy adjuvant and membrane stabilizer. These had long blood circulation times and active targeting abilities, thus creating multifunctionality of the liposomes and facilitating drug administration to the GC cells.
Methods: Three ginsenosides with different structures were used to formulate the unique nanocarrier, which was prepared using the thin-film hydration method. The stability of the ginsenoside liposomes was determined by particle size analysis using dynamic light scattering. The long circulation time of ginsenoside liposomes was compared with that of conventional liposome and polyethylene glycosylated liposomes in vivo. The active targeting effect of ginsenoside liposomes was examined with a GC xenograft model using an in vivo imaging system. To examine the antitumor activity of ginsenoside liposomes against GC, MTT, cell cycle, and apoptosis assays were performed on BGC-823 cells in vitro and PTX-loaded ginsenoside liposomes were prepared to evaluate the therapeutic efficacy on GC in vivo.
Results: The ginsenosides stabilized the liposomes in a manner similar to cholesterol. We confirmed the successful delivery of the bioactive combination drugs and internalization into GC cells via analysis of the glucose-related transporter recognition and longer blood circulation time. PTX was encapsulated in different liposomal formulations for use as a combination therapy, in which ginsenosides were found to exert their inherent anticancer activity, as well as act synergistically with PTX. The combination therapy using these targeted liposomes significantly suppressed GC tumor growth and outperformed most reported PTX formulations, including Lipusu® and Abraxane®.
Conclusion: We established novel ginsenoside-based liposomes as a tumor-targeting therapy, in which ginsenoside functioned not only as a chemotherapy adjuvant, but also as a functional membrane material. Ginsenoside-based liposomes offer a novel platform for anticancer drug delivery and may lead to a new era of nanocarrier treatments for cancer.
Keywords: Ginsenoside, liposome, multifunction, gastric cancer, combination therapy, paclitaxel.
 Global reach, higher impact
Global reach, higher impact