13.3
Impact Factor
Theranostics 2019; 9(16):4608-4623. doi:10.7150/thno.35188 This issue Cite
Research Paper
A Compound AC1Q3QWB Selectively Disrupts HOTAIR-Mediated Recruitment of PRC2 and Enhances Cancer Therapy of DZNep
1. Department of Neurosurgery, Tianjin Medical University General Hospital, Tianjin 300052, China. Laboratory of Neuro-Oncology, Tianjin Neurological Institute, Department of Neurosurgery, Tianjin Medical University General Hospital and Key Laboratory of Neurotrauma, Variation, and Regeneration, Ministry of Education and Tianjin Municipal Government, Tianjin 300052, China.
2. Department of Genetics, School of Basic Medical Sciences, Tianjin Medical University.
3. Department of Pathology, Medical College of Hebei University, Baoding, Hebei 071000, China
4. Department of Neurosurgery, the Second Affiliated Hospital of Harbin Medical University, Neuroscience Institute, Heilongjiang Academy of Medical Sciences, Harbin 150086, China.
5. Department of Medicine, Department of Surgery, the University of Oklahoma Health Sciences Center, Oklahoma City, OK, 73104, USA.
6. Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou510095, China.
*These authors contributed equally to this work as first authors.
Abstract
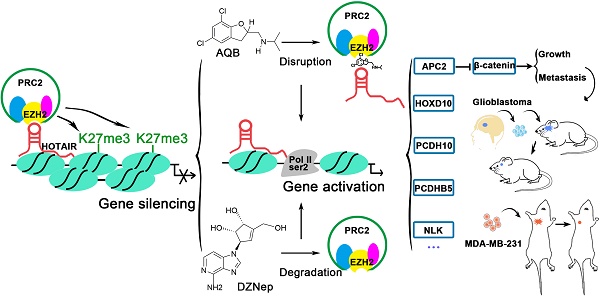
Over 20% of cancer 'driver' genes encode chromatin regulators. Long noncoding RNAs (lincRNAs), which are dysregulated in various cancers, play a critical role in chromatin dynamics and gene regulation by interacting with key epigenetic regulators. It has been previously reported that the lincRNA HOTAIR mediates recruitment of polycomb repressive complex 2 (PRC2) leading to aberrant transcriptional silencing of tumor suppressor genes in glioma and breast cancer. Thus, lincRNA HOTAIR can serve as a promising therapeutic target. Herein, we identified a small-molecule compound AC1Q3QWB (AQB) as a selective and efficient disruptor of HOTAIR-EZH2 interaction, resulting in blocking of PRC2 recruitment and increasing tumor suppressors expression.
Methods: Molecular docking and high-throughput screening were performed to identify the small compound, AQB. RIP and ChIRP assays were carried to assess the selective interference of AQB with the HOTAIR-EZH2 interaction. The effects of AQB on tumor malignancy were evaluated in a variety of cancer cell lines and orthotopic breast cancer models. The combination therapy of AQB and 3-Deazaneplanocin A (DZNep), an inhibitor of the histone methyltransferase EZH2 was used in vitro and in orthotopic breast cancer and glioblastoma patient-derived xenograft (PDX) models.
Results: Tumor cells highly expressing HOTAIR and EZH2 were sensitive to AQB. APC2, as one of the target genes, was significantly up-regulated by AQB and led to degradation of β-catenin resulting in suppression of Wnt/β-catenin signaling which may contribute to inhibition of tumor growth and metastasis in vitro and in orthotopic breast cancer models. Remarkably, AQB enhanced the toxicity of DZNep in vitro. In orthotopic breast cancer and glioblastoma patient-derived xenografts (PDX) models, the combination of low doses of AQB and DZNep realized much better killing than DZNep treatment alone.
Conclusion: AQB is a HOTAIR-EZH2 inhibitor, which blocks PRC2 recruitment and has great potential as an effective agent for targeted cancer therapy.
Keywords: HOTAIR-EZH2, DZNep, APC2, β-catenin
 Global reach, higher impact
Global reach, higher impact