13.3
Impact Factor
Theranostics 2019; 9(16):4633-4647. doi:10.7150/thno.32260 This issue Cite
Research Paper
Human Mesenchymal Stem Cell-Treated Regulatory CD23+CD43+ B Cells Alleviate Intestinal Inflammation
1. The Biotherapy Center, the Third Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510630, China.
2. Center for Stem Cell Biology and Tissue Engineering, Key Laboratory for Stem Cells and Tissue Engineering, Ministry of Education, Sun Yat-sen University, Guangzhou, 510080, China.
3. Department of Pathophysiology, Zhongshan School of Medicine, Sun Yat-sen University, Guangzhou, 510080, China.
4. Organ Transplant Center, the First Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510080, China.
5. Hematological Department of Guangdong Province People's Hospital, Guangzhou 510080, China.
6. Institute of Blood Transfusion, Guangzhou Blood Centre, Guangzhou, 510095, China
7. Department of Biochemistry, Zhongshan School of Medicine, Sun Yat-sen University, Guangzhou, 510080, China.
8. Key Laboratory of Protein Modification and Degradation, School of Basic Medical Sciences, Affiliated Cancer Hospital and Institute of Guangzhou Medical University, Guangzhou 511436, China
*Authors contributed equally to this work.
Abstract
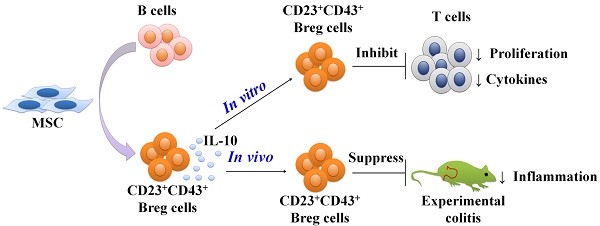
Rationale: Mesenchymal stem cells (MSCs) have been demonstrated to ameliorate inflammatory bowel disease by their actions on multiple immune cells, especially on regulatory B cells (Breg cells). However, the phenotypes and functions of human MSCs (hMSCs)-treated Breg cell subsets are not yet clear.
Methods: Purified B cells were cocultured with MSCs and the phenotypes and immunomodulatory functions of the B cells were analyzed by FACS and proliferation assays in vitro. Also, a trinitrobenzenesulfonic acid-induced mouse colitis model was employed to detect the function of MSC-treated Breg cells in vivo.
Results: We demonstrated that coculturing with hMSCs significantly enhanced the immunomodulatory activity of B cells by up-regulating IL-10 expression. We then identified that a novel regulatory B cell population characterized by CD23 and CD43 phenotypic markers could be induced by hMSCs. The CD23+CD43+ Breg cells substantially inhibited the inflammatory cytokine secretion and proliferation of T cells through an IL-10-dependent pathway. More significantly, intraperitoneal injection of hMSCs ameliorated the clinical and histopathological severity in the mouse experimental colitis model, accompanied by an increase in the number of CD23+CD43+ Breg cells. The adoptive transfer of CD23+CD43+ B cells effectively alleviated murine colitis, as compared with the CD23-CD43- B cells. Treatment with CD23+CD43+ B cells, and not hMSCs, substantially improved the symptoms of colitis in B cell-depleted mice.
Conclusion: the novel CD23+CD43+ Breg cell subset appears to be involved in the immunomodulatory function of hMSCs and sheds new light on elucidating the therapeutic mechanism of hMSCs for the treatment of inflammation-related diseases.
Keywords: mesenchymal stem cells, regulatory B cells, inflammatory bowel disease, interleukin-10
 Global reach, higher impact
Global reach, higher impact