13.3
Impact Factor
Theranostics 2019; 9(16):4648-4662. doi:10.7150/thno.35414 This issue Cite
Research Paper
Loureirin B suppresses RANKL-induced osteoclastogenesis and ovariectomized osteoporosis via attenuating NFATc1 and ROS activities
1. Department of Joint Orthopaedic, the First Affiliated Hospital, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510405, China
2. School of Biomedical Sciences, University of Western Australia, Perth, WA 6009, Australia
3. The Lab of Orthopaedics of Chinese Medicine of Lingnan Medical Research Center, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510405, China
4. The First Clinical Medical College, Guangzhou University of Chinese Medicine, Guangzhou, Guangdong 510405, China
5. Research Centre for Regenerative Medicine, Guangxi Medical University, Nanning, Guangxi 530021, China
* Yuhao Liu and Chao Wang contributed equally to this work.
Abstract
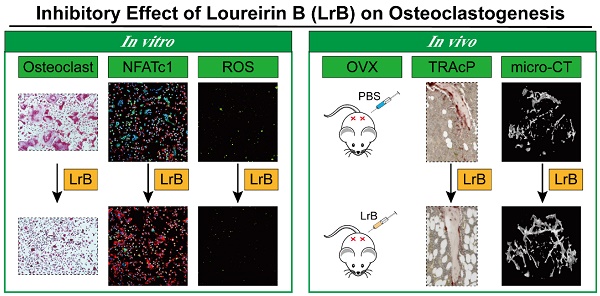
Rationale: Osteoporosis is a severe bone disorder that is a threat to our aging population. Excessive osteoclast formation and bone resorption lead to changes in trabecular bone volume and architecture, leaving the bones vulnerable to fracture. Therapeutic approaches of inhibiting osteoclastogenesis and bone resorption have been proven to be an efficient approach to prevent osteoporosis. In our study, we have demonstrated for the first time that Loureirin B (LrB) inhibits ovariectomized osteoporosis and explored its underlying mechanisms of action in vitro. Methods: We examined the effects of LrB on RANKL-induced osteoclast differentiation and bone resorption, and its impacts on RANKL-induced NFATc1 activation, calcium oscillations and reactive oxygen species (ROS) production in osteoclasts in vitro. We assessed the in vivo efficacy of LrB using an ovariectomy (OVX)-induced osteoporosis model, which was analyzed using micro-computed tomography (micro-CT) and bone histomorphometry. Results: We found that LrB represses osteoclastogenesis, bone resorption, F-actin belts formation, osteoclast specific gene expressions, ROS activity and calcium oscillations through preventing NFATc1 translocation and expression as well as affecting MAPK-NFAT signaling pathways in vitro. Our in vivo study indicated that LrB prevents OVX-induced osteoporosis and preserves bone volume by repressing osteoclast activity and function. Conclusions: Our findings confirm that LrB can attenuate osteoclast formation and OVX-induced osteoporosis. This novel and exciting discovery could pave the way for the development of LrB as a potential therapeutic treatment for osteoporosis.
Keywords: Loureirin B, Nuclear factor of activated T cells 1, Reactive oxygen species, Osteoclast, Osteoporosis
 Global reach, higher impact
Global reach, higher impact