13.3
Impact Factor
Theranostics 2019; 9(16):4663-4677. doi:10.7150/thno.34464 This issue Cite
Research Paper
Biomimetic open porous structured core-shell microtissue with enhanced mechanical properties for bottom-up bone tissue engineering
Department of Plastic Surgery, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan 430022, China.
# These authors contributed equally to this work.
Abstract
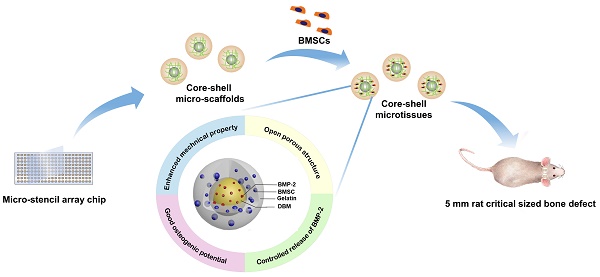
Background: Microtissues constructed with hydrogels promote cell expansion and specific differentiation by mimicking the microarchitecture of native tissues. However, the suboptimal mechanical property and osteogenic activity of microtissues fabricated by natural polymers need further improvement for bone reconstruction application. Core-shell designed structures are composed of an inner core part and an outer part shell, combining the characteristics of different materials, which improve the mechanical property of microtissues.
Methods: A micro-stencil array chip was used to fabricate an open porous core-shell micro-scaffold consisting of gelatin as shell and demineralized bone matrix particles modified with bone morphogenetic protein-2 (BMP-2) as core. Single gelatin micro-scaffold was fabricated as a control. Rat bone marrow mesenchymal stem cells (BMSCs) were seeded on the micro-scaffolds, after which they were dynamic cultured and osteo-induced in mini-capsule bioreactors to fabricate microtissues. The physical characteristics, biocompatibility, osteo-inducing and controlled release ability of the core-shell microtissue were evaluated in vitro respectively. Then microtissues were tested in vivo via ectopic implantation and orthotopic bone implantation in rat model.
Results: The Young's modulus of core-shell micro-scaffold was nearly triple that of gelatin micro-scaffold, which means the core-shell micro-scaffolds have better mechanical property. BMSCs rapidly proliferated and retained the highest viability on core-shell microtissues. The improved osteogenic potential of core-shell microtissues was evidenced by the increased calcification based on von kossa staining and osteo-relative gene expression. At 3months after transplantation, core-shell microtissue group formed the highest number of mineralized tissues in rat ectopic subcutaneous model, and displayed the largest amount of new bony tissue deposition in rat orthotopic cranial defect.
Conclusion: The novel core-shell microtissue construction strategy developed may become a promising cell delivery platform for bone regeneration.
Keywords: Core-shell, microtissue, bottom-up, demineralized bone matrix, biomimetic
 Global reach, higher impact
Global reach, higher impact