13.3
Impact Factor
Theranostics 2019; 9(16):4740-4755. doi:10.7150/thno.33520 This issue Cite
Research Paper
Employing Macrophage-Derived Microvesicle for Kidney-Targeted Delivery of Dexamethasone: An Efficient Therapeutic Strategy against Renal Inflammation and Fibrosis
1. Institute of Nephrology, Zhong Da Hospital, School of Medicine, Southeast University, Nanjing, China
2. State Key Laboratory of Bioelectronics, Jiangsu Key Laboratory for Biomaterials and Devices, School of Biological Sciences and Medical Engineering, Southeast University, Nanjing, China
3. Division of Nephrology, Department of Medicine, Duke University and Durham VA Medical Centers, Durham, North Carolina, United States
Abstract
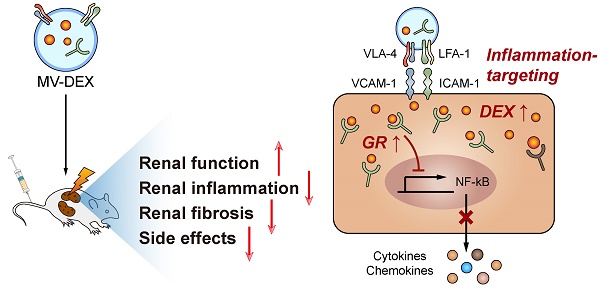
Although glucocorticoids are the mainstays in the treatment of renal diseases for decades, the dose dependent side effects have largely restricted their clinical use. Microvesicles (MVs) are small lipid-based membrane-bound particles generated by virtually all cells. Here we show that RAW 264.7 macrophage cell-derived MVs can be used as vectors to deliver dexamethasone (named as MV-DEX) targeting the inflamed kidney efficiently.
Methods: RAW macrophages were incubated with dexamethasone and then MV-DEX was isolated from the supernatants by centrifugation method. Nanoparticle tracking analysis, transmission electron microscopy, western blot and high-performance liquid chromatography were used to analyze the properties of MV-DEX. The LC-MS/MS was applied to investigate the protein compositions of MV-DEX. Based on the murine models of LPS- or Adriamycin (ADR)-induced nephropathy or in-vitro culture of glomerular endothelial cells, the inflammation-targeting characteristics and the therapeutic efficacy of MV-DEX was examined. Finally, we assessed the side effects of chronic glucocorticoid therapy in MV-DEX-treated mice.
Results: Proteomic analysis revealed distinct integrin expression patterns on the MV-DEX surface, in which the integrin αLβ2 (LFA-1) and α4β1 (VAL-4) enabled them to adhere to the inflamed kidney. Compared to free DEX treatment, equimolar doses of MV-DEX significantly attenuated renal injury with an enhanced therapeutic efficacy against renal inflammation and fibrosis in murine models of LPS- or ADR-induced nephropathy. In vitro, MV-DEX with about one-fifth of the doses of free DEX achieved significant anti-inflammatory efficacy by inhibiting NF-κB activity. Mechanistically, MV-DEX could package and deliver glucocorticoid receptors to renal cells, thereby, increasing cellular levels of the receptor and improving cell sensitivity to glucocorticoids. Notably, delivering DEX in MVs significantly reduced the side effects of chronic glucocorticoid therapy (e.g., hyperglycemia, suppression of HPA axis).
Conclusion: In summary, macrophage-derived MVs efficiently deliver DEX into the inflamed kidney and exhibit a superior capacity to suppress renal inflammation and fibrosis without apparent glucocorticoid adverse effects. Our findings demonstrate the effectiveness and security of a novel drug delivery strategy with promising clinical applications.
Keywords: renal inflammation, renal fibrosis, macrophage-derived microvesicles, dexamethasone, drug delivery
 Global reach, higher impact
Global reach, higher impact