13.3
Impact Factor
Theranostics 2019; 9(16):4811-4826. doi:10.7150/thno.35919 This issue Cite
Research Paper
Dengue and Zika Virus Domain III-Flagellin Fusion and Glycan-Masking E Antigen for Prime-Boost Immunization
1. Institute of Biotechnology, National Tsing Hua University, Hsinchu 30013, Taiwan
2. Institute of Cellular and Organismic Biology, Academia Sinica, Taipei 11529, Taiwan
3. Department of Medical Science, National Tsing Hua University, Hsinchu 30013, Taiwan
Abstract
The viral E proteins of dengue virus (DENV) and Zika virus (ZIKV) are the major viral proteins involved in receptor binding and fusion, and for the induction of protective antibodies against viral infections. DIII of the E proteins is an independent domain and stretches out on the virion surface that can elicit type-specific neutralizing antibodies. For recombinant DIII vaccine development, prime-boost immunizations can provide an advantage of eliciting more type-specific neutralizing antibodies by recalling DIII antigens after DIII booster to improve protection.
Methods: The DIII of the E genes of DENV and ZIKV were fused with bacterial fliC gene for the expression of flagellin-DIII (FliC-DIII) fusion proteins. Prime-boost immunization strategies by the second-dose booster of four DENV serotype or ZIKV FliC-DIII fusion proteins were used to investigate the induction of neutralizing antibodies and protection against viral infections. Cross-reactive non-neutralizing antibodies in each group of antisera were also examined using in vitro antibody-dependent enhancement (ADE) assay. A series of glycan-masking E antigens were finally constructed for prime-boost immunizations to abolish the elicitation of cross-reactive non-neutralizing antibodies for ADE activity.
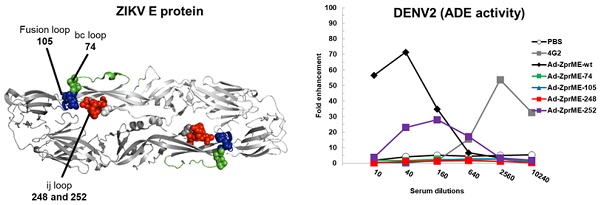
Results: We showed that inclusion of a bivalent live-attenuated vaccine with a FliC-DIII booster is superior in eliciting neutralization titers and protection in vivo against all four-serotype DENVs. We also demonstrated that recombinant adenovirus vectors encoding four-serotype DENV prMEs with a FliC-DIII prime-boost scheme is capable of eliciting good antibody responses. In contract, recombinant adenovirus vector of ZIKV prME gene priming, followed by ZIKV FliC-DIII booster did not improve vaccine efficacy. The glycan-masking mutation on the ZIKV E protein ij loop (E-248NHT), but not on DENV2 E protein ij loop (E-242NHT), resulted in abolishing the elicitation of cross-reactive antibodies for DENV and ZIKV infection enhancements.
Conclusions: Our findings can provide useful information for designing novel immunogens and vaccination strategies in an attempt to develop a safe and efficacious DENV or ZIKV vaccine.
Keywords: flagellin-domain III fusion, dengue and Zika viruses, neutralizing antibodies, cross-reactive antibodies, prime-boost vaccines
 Global reach, higher impact
Global reach, higher impact