13.3
Impact Factor
Theranostics 2019; 9(18):5227-5245. doi:10.7150/thno.34273 This issue Cite
Research Paper
LncRNA PCNAP1 modulates hepatitis B virus replication and enhances tumor growth of liver cancer
1. Department of Cancer Research, College of Life Sciences, Nankai University, Tianjin 300071, China.
2. Key Laboratory of Plant Resources and Chemistry in Arid Regions, Xinjiang Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, Urumqi 830011, P.R. China.
3. Beijing Vitalstar Biotechnology Co. Ltd.
* Present address: Department of Animal Science, Michigan State University, East Lansing, MI USA 48824.
# Jinyan Feng and Guang Yang contributed equally as joint first authors.
Abstract
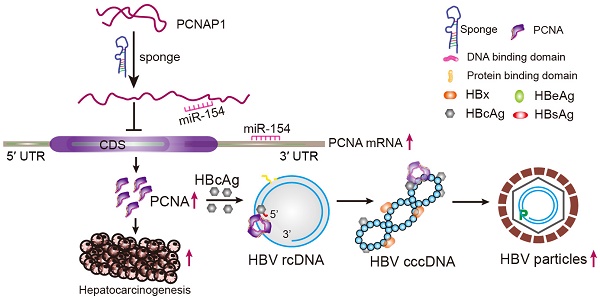
Rationale: Hepatitis B virus (HBV) is a major risk factor for liver cancer, in which HBV covalently closed circular DNA (cccDNA) plays crucial roles. However, the effect of pseudogene-derived long noncoding RNAs (lncRNAs) acting as functional regulators of their ancestral gene expression on HBV replication and hepatocellular carcinoma (HCC) remains unclear. In this study, we speculated that the pseudogene-derived lncRNA PCNAP1 and its ancestor PCNA might modulate HBV replication and promote hepatocarcinogenesis.
Methods: We investigated the roles of lncRNA PCNAP1 in contribution of HBV replication through modulating miR-154/PCNA/HBV cccDNA signaling in hepatocarcinogenesis by using CRISPR/Cas9, Southern blot analysis, confocal assays, et al. in primary human hepatocytes (PHH), HepaRG cells, HepG2-NTCP cells, hepatoma carcinoma cells, human liver-chimeric mice model, transgenetic mice model, in vitro tumorigenicity and clinical patients.
Results: Interestingly, the expression levels of PCNAP1 and PCNA were significantly elevated in the liver of HBV-infectious human liver-chimeric mice. Clinically, the mRNA levels of PCNAP1 and PCNA were increased in the liver of HBV-positive/HBV cccDNA-positive HCC patients. Mechanistically, PCNA interacted with HBV cccDNA in a HBc-dependent manner. PCNAP1 enhanced PCNA through sponging miR-154 targeting PCNA mRNA 3′UTR. Functionally, PCNAP1 or PCNA remarkably enhanced HBV replication and accelerated the growth of HCC in vitro and in vivo.
Conclusion: We conclude that lncRNA PCNAP1 enhances the HBV replication through modulating miR-154/PCNA/HBV cccDNA signaling and the PCNAP1/PCNA signaling drives the hepatocarcinogenesis. Our finding provides new insights into the mechanism by which lncRNA PCNAP1 enhances HBV replication and hepatocarcinogenesis.
Keywords: LncRNA PCNAP1, HBV, HBV cccDNA, PCNA, hepatocarcinogenesis.
 Global reach, higher impact
Global reach, higher impact