13.3
Impact Factor
Theranostics 2019; 9(18):5246-5260. doi:10.7150/thno.34713 This issue Cite
Research Paper
APLN promotes hepatocellular carcinoma through activating PI3K/Akt pathway and is a druggable target
1. Institute of Digestive Disease and Department of Medicine and Therapeutics, State Key laboratory of Digestive Disease, Li Ka Shing Institute of Health Sciences, CUHK Shenzhen Research Institute, The Chinese University of Hong Kong, Hong Kong SAR.
2. Department of Gastroenterology, The Third Affiliated Hospital of Sun Yat-Sen University, Guangzhou, 510630, China
3. Precision Medicine Institute, First Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510080, China
4. Department of Liver Surgery, First Affiliated Hospital, Sun Yat-sen University, Guangzhou, 510080, China
5. Department of Anatomical and Cellular Pathology, The Chinese University of Hong Kong, Shatin, Hong Kong, China
Abstract
Background: The pathogenesis of hepatocellular carcinoma (HCC) is a multistep process contributed by the accumulation of molecular alterations. We identified Apelin (APLN) as an outlier gene up-regulated in hepatocellular carcinoma (HCC) through RNA-Seq and microarray analysis. We aimed to investigate its function, mechanism of action and clinical implication in HCC.
Methods: Gene expression and clinical implication of APLN were assessed in multiple human HCC cohorts. Ectopic expression and silencing of APLN were performed to determine its function. The therapeutic potential of APLN and its downstream pathway was investigated using in vitro and in vivo models.
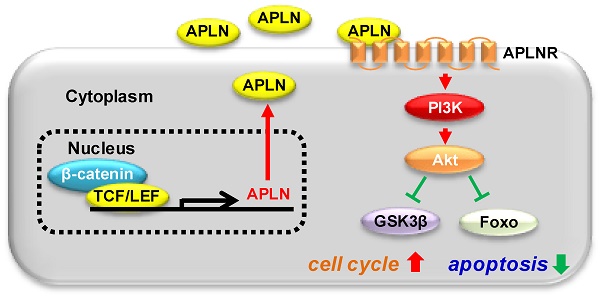
Results: APLN overexpression was commonly observed in more than 80% of HCCs and independently predicted poorer survival of patients in three independent HCC cohorts. Apelin up-regulation was mediated by active β-catenin, which binds to the APLN promoter to induce transcription. Ectopic APLN expression in HCC cells promoted cell proliferation, accelerated G1/S progression and inhibited apoptosis, whilst APLN knockdown exerted opposite effects in vitro and inhibited HCC xenograft growth in mice. Mechanistically, APLN activated phosphatidylinositol 3-kinase/protein kinase B (PI3K/Akt) pathway via APLN receptor, leading to increased expression of phospho-glycogen synthase kinase 3β (p-GSK3β) and cyclin D1. Pharmacological targeting of APLN by ML221 was safe and effective in inhibiting APLN-PI3K/Akt cascade and HCC growth in vitro and in vivo.
Conclusions: Our findings unraveled an oncogenic role of APLN in HCC, and that targeting of APLN might be a promising for HCC treatment. APLN may serve as an independent prognostic factor for HCC patients.
Keywords: Hepatocellular carcinoma, APLN, prognosis, PI3K/Akt.
 Global reach, higher impact
Global reach, higher impact