13.3
Impact Factor
Theranostics 2019; 9(18):5261-5281. doi:10.7150/thno.33183 This issue Cite
Research Paper
Focused ultrasound-augmented targeting delivery of nanosonosensitizers from homogenous exosomes for enhanced sonodynamic cancer therapy
Key Laboratory of Medicinal Resources and Natural Pharmaceutical Chemistry, Ministry of Education, National Engineering Laboratory for Resource Developing of Endangered Chinese Crude Drugs in Northwest of China, College of Life Sciences, Shaanxi Normal University, Xi'an, Shaanxi, China.
*These authors contributed equally to this work.
Abstract
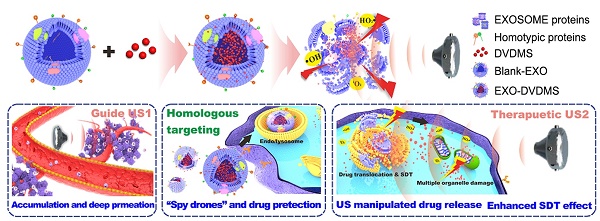
Sonodynamic therapy (SDT), wherein focused ultrasound is used to guide the site-specific delivery of nano-sonosensitizers and trigger profound sono-damage, has great potential in cancer theranostics. The development of nanosensitizers with high sono-activatable efficiency and good biosafety is however challenging.
Methods: In this study, we designed a functionalized smart nanosonosensitizer (EXO-DVDMS) by loading sinoporphyrin sodium (DVDMS), an excellent porphyrin sensitizer with both potential therapeutic and imaging applications, onto homotypic tumor cell-derived exosomes. Because of the high binding-affinity between DVDMS and proteins, coincubation of DVDMS and exosome would result in DVDMS attached on the surface or loaded in the core of exosomes. The prepared EXO-DVDMS was applied for ultrasound-responsive controlled release and enhanced SDT.
Results: Tumor cell-derived exosomes exhibited high stability and specificity towards the homotypic tumors, along with highly controlled ultrasound-responsive drug release, and boosted reactive oxygen species (ROS) generation to augment SDT. Intriguingly, EXO-DVDMS was endocytosed by lysosomes, and the low pH in the latter triggered DVDMS relocation synergistically with the ultrasound, thereby initiating multiple cell death-signaling pathways. Furthermore, the exosomal formulation served as a functionalized nanostructure, and facilitated simultaneous imaging and tumor metastasis inhibition, that were respectively 3-folds and 10-folds higher than that of free form.
Conclusions: Taken together, our findings suggest that an extracorporeal ultrasound device can non-invasively enhance homogenous tumor targeting and SDT toxicity of EXO-DVDMS, and the developed endogenous nano-sonosensitizer is a promising nanoplatform for activated cancer theranostics.
Keywords: Nanosonosensitizer, Sinoporphyrin sodium, Exosome, Ultrasound-responsive drug release, Sonodynamic therapy
 Global reach, higher impact
Global reach, higher impact